The issues related to haemostatic disorders and their treatment are being widely discussed in the literature concerning the diagnosis and management of patients diagnosed with severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) [1–3]. The findings of numerous worldwide studies have demonstrated that the haemostatic system is one of the most important systems substantially impaired by the viral infection, which can result in serious life-threatening complications [4–6]. It is already known that hypercoagulation [7, 8] is the dominant coagulopathy in patients infected with SARS-CoV-2. Its consequences in the form of blood clots and emboli in the blood vessels of various organs in combination with thrombocytopathy lead to severe disorders of haemostasis in some patients and require ICU treatment [9–11]. According to the literature data, the incidence of thromboembolic incidents in this group of patients reaches 43% and increases with the length of hospitalization [12, 13]. The incidence of pulmonary embolism, which is observed most frequently, is six-fold higher in patients with COVID-19-associated acute respiratory distress syndrome (ARDS), as compared to patients with ARDS of some different aetiology [14]. Therefore, optimal diagnosis of haemostatic disorders and effective treatment of coagulopathy are the elements of therapeutic management, which can improve the prognosis in most critically ill patients.
The aim of the paper was to synthesise information on the diagnosis and therapy of haemostatic disorders in patients infected with SARS-CoV-2 requiring intensive care. The authors did not present their own recommendations or guidelines, but merely attempted to summerise the existing knowledge on the subject in an orderly manner.
EPIDEMIOLOGY OF COVID-19 IN POLAND AND WORLDWIDE
In March 2020, the World Health Organization declared COVID-19 a pandemic caused by a new type of coronavirus: SARS-CoV-2. During the infection, patients develop pneumonia, the main symptoms of which are fever, dry cough, and progressive shortness of breath. The first reports of pneumonia cases emerged from the Chinese City of Wuhan, Hubei Province, in December 2019. In Poland, the first case was confirmed on March 4, 2020 in an individual returning from abroad. At the beginning of December 2020, the number of infections worldwide exceeded 66 million and more than 1.5 million people died; in Poland, the number of infections exceeded one million cases and 19 thousand deaths were reported [15].
PATHOPHYSIOLOGY AND PATHOMORPHOLOGY
In the pathogenesis of thromboembolic complications in COVID-19, all elements of the Virchow`s triad are involved: endothelial damage, coagulation disorders and blood flow abnormalities.
The invasion of SARS-CoV-2 into the endothelial cells leads to their activation, which initiates local inflammation. This promotes increased vascular permeability while disturbed production of nitric oxide and prostacyclin I impairs their antithrombotic properties. Upon activation, neutrophils can release neutrophil extracellular traps (NETs), which may promote thrombosis [16]. Moreover, endothelial activation results in enhanced expression of plasminogen activator inhibitor-1 (PAI-1), tissue factor (TF) and release of von Willebrand factor (vWF). The activity of thrombomodulin and tissue plasminogen activator (t-PA) decreases [17]. The third element of Virchow’s triad is abnormal blood flow. In ICU patients, this abnormal flow is caused by immobilization and angiopathies resulting from the exacerbation of chronic cardiovascular diseases, diabetes, renal failure, and age-related fragility.
The problems associated with impaired organ perfusion resulting from the presence of thromboembolic lesions have been described by Shah et al. [12]. In their study group, numerous thromboembolic complications have been observed, such as strokes, myocardial infarction, and ischaemic intestinal lesions. The authors have also reported extracorporeal circuit thrombosis in more than 12% of patients.
Anatomopathological studies of the lungs of patients who died of COVID-19 have shown massive endothelial damage and diffuse alveolar damage (DAD) in 67% of subjects [18]. Intra-alveolar fibrin deposition and the presence of vitreous membranes have been observed. Diffuse thrombotic lesions and microthrombi in the pulmonary capillaries have also been found, whose incidence was 9 times higher than in patients with influenza virus infection [10]. Furthermore, microthrombi have been detected in the arteries 1–2 mm in diameter, without complete occlusion of their lumen. The above histological findings have been confirmed by microtomographic (micro-CT 3D) studies of the lungs, which demonstrate almost complete obstruction of pre- and post-capillary vessels. Moreover, increased angiogenesis has been observed and its incidence was almost three-fold higher, as compared to patients with influenza. In autopsy studies, Wichmann et al. [18] have found deep vein thrombosis in 58% of patients. Pulmonary embolism was the direct cause of death of 1/3 of patients in the studied group. Features of disseminated intravascular coagulation (DIC) and small fibrin clots have been demonstrated in the renal glomeruli of COVID-19 patients [19].
CHARACTERISTICS OF COVID-19-ASSOCIATED COAGULOPATHY
COVID-19-associated coagulopathy (CAC) is chara-cterised by hypercoagulation [20], which is a significant factor worsening prognosis [3].
Laboratory tests in CAC demonstrate a slight increase in prothrombin time (PT) and activated partial thromboplastin time (APTT), high D-dimer levels and elevated levels of fibrinogen, factor VIII and vWF [21].
The coagulation disorders described in CAC differ from those in coagulopathies commonly occurring in ICUs, such as DIC or sepsis-induced coagulopathy (SIC). The characteristics of these three types of coagulopathy and the scales enabling their description were presented in the updated recommendations of the Agency for Health Technology Assessment and Tariffication (AHTAT) of 27.11.2020 [22]. The main differentiating features of CAC and DIC were described in Table 1.
TABLE 1
Characteristics of COVID-19-associated coagulopathy (CAC) and disseminated intravascular coagulation (DIC)
| Factor | CAC | DIC |
|---|---|---|
| Dominant picture of coagulopathy | Thrombosis | Haemorrhage |
| D-dimer | ↑ | ↑ |
| Fibrinogen | ↑ | ↓ |
| Platelets | ↓/N | ↓ |
| PT | N/↑ | ↑ |
| APTT | N/↑ | ↑ |
| Fibrinolysis | ↓ | ↑ |
Coagulation tests
D-dimer
Increased D-dimer levels are observed during COVID-19 infection. Han et al. [23] have found higher D-dimer values in the study group of 94 patients with confirmed SARS-CoV-2 infection, as compared to the control group of 40 healthy volunteers. They also observed that higher values correlated with a heavier course of COVID-19. This also applies to critically ill patients hospitalized in ICUs. High D-dimer values are one of the factors worsening the prognosis in COVID-19 [9]. The above observation has been confirmed by Tang et al. [3], who have found significantly higher values of this marker in non-survivors. A prospective observational study on the Polish population of patients treated in ICUs has demonstrated significantly higher D-dimer levels in patients who developed acute respiratory failure with PaO2/FiO2 ≤ 300 mm Hg, as compared to those who did not meet the criteria of the Berlin definition of ARDS [24].
The literature reports different D-dimer cut-off values above which the risk of death increases significantly. For instance, ≥ 2000 ng mL-1 according to Shang et al. [25] or a two times lower value according to to Shou et al. [9]. In the AHTAT recommendations, a 6-fold increase in D-dimer above the upper limit of normal (500 ng mL-1), i.e. 3000 ng mL-1, is considered relevant [22].
Fibrinogen
Fibrinogen is a coagulation factor that is involved in the final phase of clot formation after transformation into fibrin and binding to the platelets to form the final clot stabilized by factor XIII. At the same time, it is an acute phase protein produced in the liver; therefore, its serum concentration significantly increases during inflammatory processes [26]. High concentrations of fibrinogen are one of the factors worsening the prognosis in COVID-19 due to a high risk of exacerbated thromboembolic complications [27]. On the other hand, significantly reduced fibrinogen concentrations have been observed in critically ill patients in whom this coagulation factor was substantially used in the final stage of the disease, most likely due to DIC [3].
Platelets
In COVID-19 infections, both thrombocytopenia and normal platelet counts are observed. In a meta-analysis conducted by Lippi et al. [28], low platelet counts have been found in patients with more severe infections. In a retrospective study involving 191 patients with COVID-19, Shou et al. [9] have found significantly lower platelet counts in non-survivors, as compared to survivors. Despite this difference, the number of thrombocytes in both groups did not differ significantly from the reference values, and severe thrombocytopenia occurred sporadically.
Liu et al. [29] have confirmed the importance of reduced thrombocyte counts as an independent risk factor for death in severe COVID-19 cases. According to them, thrombocytopenia was associated with a 3-fold higher risk of death compared to patients with normal platelet counts. A 50 G L–1 increase in platelet counts was associated with a 40% reduction in the risk of death.
Coagulation times
The coagulation tests in the group of further 183 patients admitted to hospital with SARS-CoV-2 have shown normal or slightly prolonged coagulation times. Prolonged PT was observed more frequently in patients hospitalized in ICUs and in non-survivors [3]. This observation has been confirmed by a subsequent meta-analysis involving 6,320 patients with COVID-19. Prolongation of PT and more frequently reported APTT within the reference values may indicate that haemostatic disorders in SARS-CoV-2 infections are mainly associated with the extrinsic coagulation pathway [30].
Global tests of haemostasis
Thromboelastometry (ROTEM) and thromboelastography (TEG) are the diagnostic methods known as global tests of haemostasis (GTH). They enable quick and bedside assessment of the dynamics of formation, physical properties and lysis of a blood clot. Moreover, GTH findings allow to diagnose hypercoagulation and fibrinolysis, which is not possible in standard coagulation tests [31]. However, there are no explicit study results indicating that these methods should be routinely used for monitoring the anticoagulation therapy in patients with COVID-19.
Global tests of haemostasis – abnormal clot formation
In ROTEM studies performed in patients with SARS-CoV-2, Spiezia et al. [32] have demonstrated reduced times of clot formation and higher maximum clot firmness (MCF), indicative of a hypercoagulable state. Similar observations have been reported by Almskog et al. [33] in their prospective observational study with the control group of healthy individuals. Likewise, Pavoni et al. [34], who studied critically ill patients with COVID-19 hospitalised in ICUs, have found that ROTEM results indicated increased coagulation in more than 50% of patients. The above studies evidence that GTH can be used during the SARS-CoV-2 pandemic as a diagnostic tool to quickly identify patients with coagulopathy that increases the risk of thromboembolic complications.
Global tests of haemostasis – abnormal fibrinolysis, suppressed fibrinolysis
A disorder of fibrinolysis, particularly suppres-sed fibrinolysis, is a risk factor for thrombotic com-pli-cations, multiple organ failure and increas-ed mortality [35–37]. Therefore, the effects of this pheno-menon on haemostasis disorders in patients with COVID-19 are being studied. Tang et al. [3] have described the coagulation disorders in 449 patients with severe COVID-19 and concluded that suppressed fibrinolysis together with excessive thrombin generation may be the cause of hypercoagulation. Furthermore, on TEG testing, Wright et al. [38] have observed a complete lack of lysis at 30 minutes in 57% of patients with COVID-19, suggestive of suppressed fibrinolysis. Similar findings have been reported by Bakchoul et al. [39].
Further studies should evaluate the activators and inhibitors of fibrinolysis and plasmin-antiplasmin complexes to fully characterize the dynamics of fibrinolysis in patients with COVID-19. This may enable to assess more comprehensively whether the fibrinolysis disorders demonstrated in GTH are confirmed by the activity of proteins involved in this process and which of its elements has a significant effect on its occurrence.
Anticoagulant therapy in COVID-19 patients treated in ICU
There is a consensus on an increased risk of thromboembolic events in patients with COVID-19,which concerns 20–43% of those treated in ICUs. Such events occur mainly in the form of vein thrombosis of the lower extremities and pulmonary embolism, even in the presence of anticoagulant prophylaxis [40, 41]. Therefore, clinical trials are underway in many centres around the world to determine the most optimal anticoagulation regimen.
Choosing the optimal form of anticoagulation
The recommendations and guidelines with a wide impact can be found in the publications prepared by the International Society for Thrombosis and Haemostasis (ISTH), National Institutes of Health (NIH), Anticoagulation Forum (ACF), American Society of Hematology (ASH), American Heart Association (AHA), American College of Cardiology (ACC), European Society of Cardiology (ESC) [42–47]. The analysis of the above documents reveals that there is no single, universal, widely accepted and recommended algorithm of anticoagulant therapy. Based on them, however, the key clinical issues can be identified to minimise the risk of thromboembolic complications during ICU treatment of COVID-19 patients.
They include:
determination of the optimal dose of anticoagulant therapy (prophylactic, intermediate or therapeutic dose),
determination of the most beneficial route of administration (subcutaneous delivery or continuous intravenous infusion),
determination of the factors affecting the need to modify the dose,
determination of the most effective method for monitoring the treatment with heparins.
It is common practice in anticoagulant management of patients hospitalised in ICUs to use prophylactic doses of low-molecular-weight heparins (LMWHs).
The generally accepted contraindications for this kind of management are severe thrombocytopaenia (< 25 G L–1), active bleeding or a high risk of bleeding at the platelet count < 50 G L–1 [47–49].
In the ASH and AHA guidelines for the general population of critically ill patients, there is a strong recommendation for the use of LMWHs, which are preferred over unfractionated heparins (UFHs) and for mechanical forms of thromboprophylaxis [50, 51]. This has been confirmed by the results of the 2011 PROTECT study, demonstrating that patients receiving LMWHs have fewer pulmonary embolism incidents than the UFH group. However, no differences in the incidence of deep vein thrombosis have been found [52].
Tang et al. [53] have confirmed the efficacy of LMWHs in COVID-19 patients with high levels of D-dimer (≥ 6 times the upper limit of normal) or the SIC score ≥ 4. These patients were characterised by lower 28-day mortality, as compared to patients without thromboprophylaxis.
On April 21, 2020, the ESC presented its statement on the care of patients with cardiac conditions during the pandemic. In cases of acute pulmonary embolism, the existing guidelines should be followed. As far as COVID-19 patients are concerned, the lack of significant interactions between heparins and drugs used in experimental therapies is highlighted. Caution is recommended when non-vitamin K antagonist oral anticoagulants (NOACs) are used. Interactions with some specific antiviral agents (i.e. lopinavir and ritonavir) that increase the risk of bleeding are possible [54, 55]. Therefore, the vast majority of guidelines do not recommend the use of NOACs in these patients in ICUs; instead, the therapy with heparins is advocated. As recommended by ISTH, NOAC therapy should be considered after discharge from the ICU [47].
The only document in force in Poland providing comprehensive diagnostic and therapeutic recommendations for the care of patients with COVID-19, which was commissioned by the Minister of Health, is the guidelines of the Agency for Medical Technology Assessment and Tariffication (AHTAT) of 25.04.2020. The part on prophylactic anticoagulation recommends the routine use of heparins in prophylactic doses in all severely ill patients [56]. The document was updated on November 27, 2020 [22].
According to the comparative analysis of the European and American guidelines and recommendations published in Critical Care by Flaczyk et al. [57], the following should be considered to determine detailed indications and to optimise heparin dosages in anticoagulant prophylaxis in COVID-19 patients:
The above-mentioned analysis and updated AHTAT recommendations introduce the term of the intermediate dose of low-molecular-weight heparin. Despite an increased risk of thromboembolic complications, the routine use of therapeutic doses of heparins for anticoagulant prophylaxis is not recommended. The risks associated with higher doses of LMWHs are also highlighted by Longhitano et al. [58]. In a prospective observational study involving 74 patients, the authors have not observed reduced incidences of thromboembolic episodes in patients receiving intermediate and therapeutic doses; how-ever, they have found higher incidences of severe haemorrhagic complications in this group of patients. High incidences of thromboembolic events despite the use of therapeutic doses have also been demonstrated by Llitjos et al. [8]. However, the number of complications reported was significantly lower than in the group receiving prophylactic doses of LMWHs. The most conservative approach has been presented by the NIH, which allows any changes to the standard anticoagulation regimen only during clinical trials [59].
The synthesis of the above recommendations was graphically shown in Figure 1 [43, 46, 48, 57, 60].
FIGURE 1
Recommended modifications of anticoagulant prophylaxis in ICU depending on the clinical situation (based on [43, 46, 48, 57, 60])
In November 2020, the recommendations for the care of COVID-19 patients requiring hospitalisation were published on the Ministry of Health’s Website as part of Strategy 3.0 [61]. In the section on anticoagulant prophylaxis and treatment, the authors confirm that intermediate doses of LMWHs are appropriate for patients with severe COVID-19 with D-dimer concentrations of ≥ 1500 ng mL–1 and a Padua score of ≥ 3 points. Higher doses of LMWHs should be considered for obese patients. However, there are no detailed data concerning the adaption of anticoagulant therapy in cases of acute renal failure requiring continuous renal replacement therapy, thrombocytopaenia or extreme body weight, which are common in ICUs.
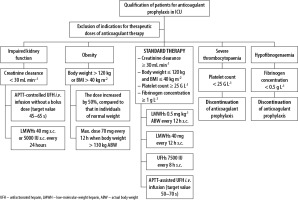
Based on the information found in Polish and international recommendations and guidelines, supplemented by the latest recommendations of the AHTAT of 27 November 2020, the authors suggest the following anticoagulant regimen in patients with COVID-19 treated in ICUs (Figure 2) [22, 57, 62, 63].
Monitoring of anticoagulant therapy (APTT, anti-Xa)
The commonly accepted practice is to determine the APTT in patients treated with UFH infusions. This form of anticoagulant therapy is particularly recommended in COVID-19 patients with a high risk of prothrombotic complications, end-stage renal failure and obesity [63].
Standard anticoagulant prophylaxis with LMWHs does not require routine monitoring. On the other hand, the use of intermediate and therapeutic doses, particularly in patients with extreme body weight, when there is an increased risk of bleeding, haematological disorders or suspected heparin resistance, should be monitored [62, 64].
The optimal method for assessing the effectiveness of LMWH therapy is anti-Xa testing [65, 66], which enables highly desirable individualisation of anticoagulant therapy; such individualisation is particularly important in anticoagulant management of COVID-19 patients. However, an adequate range of anti-X values may prove to be an important issue in clinical practice. Based on the results in the group of COVID-19 patients receiving LMWH thromboprophylaxis, Dutt et al. [67] have demonstrated low anti-Xa activity (< 0.1 IU mL–1) in 95% of patients treated in ICUs compared with 27% of patients not requiring intensive care. In view of the above, some authors indicate that determinations of anti-Xa activity are also grounded when prophylactic doses are used in COVID-19 patients, with a target value of 0.2–0.4/0.5 IU mL–1 [68]. Due to the reports demonstrating insufficient anti-Xa activity when prophylactic doses are used, higher (intermediate or therapeutic) doses of heparins have been considered to achieve the expected anti-Xa values in COVID-19 patients treated in ICUs [13, 41].
Antiplatelet therapy
According to the management regimen in some centres, LMWH or UFH treatment should be supplemented with antiplatelet drugs, the most common of which is acetylsalicylic acid.
The results of a retrospective analysis carried out by Chow et al. [69] in a group of 412 patients, have demonstrated that the use of ASA reduces the need for mechanical ventilation, the number of ICU admissions and intra-hospital mortality. However, it does not affect the incidence of severe haemorrhages and thromboembolic episodes.
At present, the guidelines and recommendations do not advocate routine antiplatelet therapy unless it has been chronically used before hospitalisation due to COVID-19 [22, 59].
Conclusions
The current findings on COVID-19 indicate that the disease is strongly associated with haemostasis disorders, resulting in hypercoagulation. The clinical picture of this coagulopathy in the form of respiratory failure and frequent dysfunctions of other systems is the result of micro- and macro-emboli. In this context, the implementation of optimal prevention or anticoagulant treatment is essential for the care of COVID-19 patients. Patients infected with SARS-CoV-2 with extreme body weight or hospitalized in ICUs require special attention during prevention or treatment with heparins. The above synthesis of information from current guidelines and recommendations allows to conclude that LMWH doses should be individualised in these groups of patients considering the results of anti-Xa activity monitoring; when UFHs are used, anti-Xa or APTT should be determined. However, the knowledge of all pathophysiological processes resulting from SARS-CoV-2 infection and the host immune response is incomplete and requires further research. We hope that the heparin dosing algorithm for ICU patients presented by us and based on the most up-to-date information will prove useful for optimizing anticoagulant therapy.



