Introduction
Dupilumab is a fully human monoclonal antibody [1, 2] that blocks the shared component of interleukin 4 (IL-4) and IL-13 receptors, which are key factors in type 2 inflammation in multiple diseases, thus inhibiting their signalling [3]. Various atopic or allergic diseases are included in type 2 inflammatory diseases, such as atopic dermatitis (AD), asthma, allergic rhinitis, and chronic rhinitis with nasal polyps (CRSwNP). Since its approval for the treatment of moderate-to-severe AD in 2017 by the US Food and Drug Administration (FDA), dupilumab has become widely used, with approval for additional indications [4] in different countries.
Eotaxin-3/C-C motif chemokine ligand 26 (CCL26), a member of the CC family of chemokines, exerts its chemotactic activity on eosinophils and basophils as target cells [5]. Its production is mediated by Th2 response mediators, such as IL-4 and IL-13, through the activation of the transcription factor STAT-6 [6]. Eotaxin-3 is a potent eosinophil attractant [5] and can induce eosinophil recruitment and activation in the airways of patients with asthma. Previous studies have also shown that serum levels of eotaxin-3 have a notable correlation with the disease activity of AD [7].
Pulmonary and activated regulated chemokine (PARC)/CCL18, a small protein derived from alveolar macrophages that acts as a chemo-attractant, is mainly secreted by antigen-presenting cells (APCs), eosinophils [8], and mast cells after FcεRI cross-linking. Its production is induced by microbial product, pathogen, and allergen exposure both in vitro and in vivo [9] and is further regulated by inflammatory cytokines; that is, PARC is upregulated in vitro by IL-4, IL-13, and IL-10 and downregulated by interferon γ (IFN-γ) and hypoxia [10].
Previous studies have shown that serum biomarkers of PARC and eosinophil-activated chemokines (including eotaxin-3) decreased significantly after dupilumab treatment [11–13]. However, these studies varied in terms of the disease type, number of patients, type of study, country or region, and time point of evaluation. The statistical power of individual studies is limited. Such factors lead to inconsistent results. As potent and easily overlooked biomarkers in type 2 inflammatory diseases, both require comprehensive analyses.
Aim
In this study, we evaluated the changes in eotaxin-3 and PARC levels after treatment with dupilumab through a meta-analysis, aiming to provide more precise and comprehensive results.
Material and methods
Literature search
Five databases, including PubMed, Web of Science, EMBASE, OVID, and Scopus, were searched from their establishment date to 2023.02.20. The search strategies varied for different databases and are described in the Supplemental Material.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) studies on the effect of dupilumab in humans; (2) patients with type 2 inflammatory diseases eligible for the use of dupilumab; (3) levels of eotaxin-3 or PARC before treatment were present in or could be extracted from the publications; (4) the dose of dupilumab and the time point of evaluation were shown; and (5) levels of eotaxin-3 or PARC after treatment were present in or could be extracted from the publications.
The exclusion criteria were as follows: (1) conference publications, case reports, review articles, commentary articles or animal studies; (2) overlapping data; (3) before or after treatment, insufficient information was provided to calculate or extract the level of eotaxin-3 or PARC to mean ± SD form; (4) the time point of evaluation after treatment was not clear; and (6) dosing data could not be extracted separately if two or more doses were applied in a group.
Two investigators (Leyi Wang and Boyang Zhou) completed the procedure separately. In cases of disagreement, another investigator (Haiyan Cheng) checked the procedure and made the final decision.
Data extraction
After studies were included, the following information was extracted: (1) study ID: first author and year of publication; (2) diseases of included patients; (3) study quality; (4) country where the study was conducted; (5) patient age; (6) administered dose; (7) unit of eotaxin-3 or PARC; (8) information before treatment: the number of patients and the eotaxin-3 or PARC level; (9) information after treatment: weeks of assessment, the number of patients and the eotaxin-3 or PARC level.
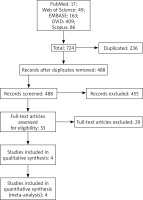
Two investigators (Leyi Wang and Boyang Zhou) completed the data extraction. Another investigator (Linfeng Li) checked and confirmed the information (Figure 1).
Methodological quality assessment
Two investigators (Leyi Wang and Boyang Zhou) conducted the assessment of study quality. For randomized studies, the Cochrane risk of bias tool was used. The Cochrane risk of bias tool consists of the evaluation of selection bias, performance bias, detection bias, attrition bias, reporting bias, and other bias. Within each bias evaluation, there are three levels, including low risk, unclear risk, and high risk. The figures can be generated in Review Manager 5.3 software. For non-randomized studies, the Newcastle-Ottawa Scale (NOS) was used [14]. The NOS consists of scores for selection, comparability, and exposure. With a total score of 9, studies scoring ≥ 6 were considered high quality. If any controversy was met, another investigator (Linfeng Li) checked the evaluation procedure and made the decision.
Statistical analysis
The standardized mean difference (SMD) was used to evaluate the change, aiming to eliminate the impact of measurement differences. A p-value < 0.05 was defined as significant. If the data were shown only in the figures, the software GetData Graph Digitizer was used to obtain the numerical values. If the data in the study were not in the form of mean ± SD, transformations were conducted (from https://www.math.hkbu.edu.hk/~tongt/papers/median2mean.html).
In data combination, fixed- or random-effect models were used based on the between-study heterogeneity evaluated from the Q-statistical test and I2 test. In each comparison, if I2 > 50% and p-value < 0.10, the heterogeneity was significant, and a random-effect model was used; otherwise, a fixed-effect model was used.
In each comparison, if two or more studies were included, it was necessary to test the impact of each individual study by means of a sensitivity analysis, in which each study was individually omitted. For each comparison, funnel plots were required to estimate publication bias if five or more studies were included. All the analysis procedures were performed in Review Manager 5.3 software.
Results
Selection of the studies
A total of 724 publications were obtained from all the sources. Of these, 236 were duplicates, and 488 studies were first included for title and abstract screening. In this selection, 455 studies were then excluded, and 33 full-text articles were screened. After thorough screening according to the inclusion and exclusion criteria, four studies were included and eligible for the meta-analysis. The selection process is shown in Figure 1.
Basic information of the included studies
In total, four studies were included in our meta-analysis [11–14]. Three studies were randomized controlled trials (RCTs) [12–14], and one was a prospective observational cohort study [11]. These studies were conducted in the United States and the Netherlands, respectively, and one international RCT study was conducted in North America, Europe, Asia, Australia, and South America [12]. Asthma was the major disease investigated in two studies [12, 13]. In addition, one study investigated atopic dermatitis (AD) [11], one investigated chronic rhinosinusitis with nasal polyps (CRSwNP) [12], and one investigated coronavirus disease 2019 (COVID-19) [14]. Doses also varied in these studies. A dose of 300 mg q2w was administered in 3 studies [11, 12, 14], which was the most. Additionally, a dose of 300 mg qw was administered in one study [13]. Before treatment, a total of 877 patients were included in eotaxin-3-related studies. The evaluation time points were week 1, week 2, week 4, week 8, week 12, week 16, week 24, week 36 and week 52. After treatment, the number of patients varied from 19 to 734 at each time point (Table 1). Thirty-five patients were included in the PARC-related study before and after treatment. The evaluation time points were week 4, week 8, week 12 and week 16.
Table 1
Characteristics and information of the included studies
Assessment of study quality
For RCT studies, study quality assessment was performed in Review Manager 5.3 software through the Cochrane risk of bias tool. These studies were at low risk (green) and unclear risk (yellow) of bias. For observational studies, study quality was assessed through the Newcastle-Ottawa Scale (NOS). The only observational study had a NOS score of 9. All included studies were considered to be of high quality (Supplementary Figure S1).
Results for eotaxin-3 level changes after treatment
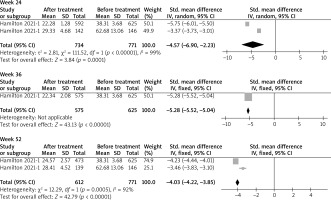
At week 1, a total of 71 patients before treatment and 69 patients after treatment were included. The level of eotaxin-3 was not seen significantly decreased, with SMD = –0.39 (95% CI: –1.78, 0.99) (p > 0.05). Similar results were presented at week 12, with SMD = –2.60 (95% CI: –5.77, 0.57) (p > 0.05), based on data from 712 patients before treatment and 702 patients after treatment. At weeks 2, 8 and 16, a total of 19, 87 and 35 patients before and after treatment were included, respectively. The pooled data showed a significant decrease in eotaxin-3 levels, with SMD = –0.94 (95% CI: –1.61, –0.27); –1.20 (95% CI: –1.52, –0.88); and –1.31 (95% CI: –1.83, –0.79) (p < 0.05), respectively. At week 4, a total of 87 patients before treatment and 84 patients after treatment were included. The pooled data showed a significant decrease in eotaxin-3 levels, with SMD = –1.17 (95% CI: –1.49, –0.84) (p < 0.05). At week 24, a total of 771 patients before treatment and 734 patients after treatment were included. The pooled data showed a significant decrease in eotaxin-3 levels, with SMD = –4.57 (95% CI: –6.90, –2.33) (p < 0.05). At week 36, a total of 625 patients before treatment and 575 patients after treatment were included. The pooled data showed a significant decrease in eotaxin-3 levels, with SMD = –5.28 (95% CI: –5.52, –5.04) (p < 0.05). At week 52, a total of 771 patients before treatment and 612 patients after treatment were included. The pooled data showed a significant decrease in eotaxin-3 levels, with SMD = –4.03 (95% CI: –4.22, –3.85) (p < 0.05). The results are shown in Figure 2.
Results for PARC level changes after treatment
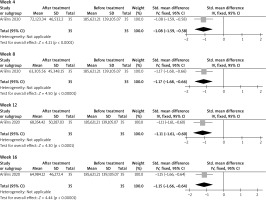
A total of 35 patients were included before and after treatment. The pooled data showed that PARC levels decreased significantly at weeks 4, 8, 12 and 16, with SMD = –1.08 (95% CI: –1.59, –0.58); –1.17 (95% CI: –1.68, –0.66); –1.11 (95% CI: –1.61, –0.60); and –1.15 (95% CI: –1.66, –0.64) (p < 0.05), respectively. The results are shown in Figure 3.
Results of the sensitivity analysis
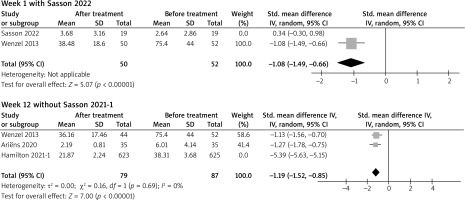
Sensitivity analysis was conducted for each comparison with two or more studies included. Robust results were suggested at weeks 2, 4, 8, 16, 24, 36 and 52, as no significant changes were shown after omitting each study. At week 1, the difference became statistically insignificant without one RCT study (Sasson 2022). At week 12, two study groups from one study (Hamilton 2021) were included. When one study group was omitted (Hamilton 2021), the difference became statistically insignificant (Figure 4).
Discussion
Currently, several clinical studies on dupilumab have been published, including LIBERTY AD SOLO 1 (NCT02277743) and SOLO 2 (NCT02277769) [15], LIBERTY AD CHRONOS (NCT02260986) [16], LIBERTY AD ADOL (NCT03054428) [17], and LIBERTY AD OLE (NCT01949311) [18]. In these studies, the primary objective was efficacy assessment rather than biomarker measurement. However, eotaxin-3 and PARC play an important role in diagnosis, classification, efficacy assessment, and prognosis. Therefore, further attention is needed.
Evidence from laboratory and clinical studies found that eotaxin-3 and PARC levels were reduced in patients treated with dupilumab. However, for individual studies, the statistical power is often limited due to the number of patients, time points of assessment, etc. Therefore, studies with more extensive analysis are needed. Through meta-analyses, data from multiple studies are integrated to provide stronger statistical power. In this study, we focused on the changes in eotaxin-3 and PARC levels after dupilumab treatment for different type 2 inflammatory diseases. To our knowledge, this is the first meta-analysis focusing on changes in eotaxin-3 and PARC levels after dupilumab treatment.
After a comprehensive literature search and data extraction, four studies were finally included. Analysing all the data in the literature together, we found that the level of eotaxin-3 did not decrease significantly at week 1, with SMD = –0.39 (95% CI: –1.78, 0.99). Afterwards, at weeks 2, 4, 8, 16, 24, 36, and 52, the level of eotaxin-3 decreased significantly. However, we found that the decrease at week 12 was not statistically significant. We checked the data and results again, after which we believed that the insignificance was due to the random-effect model we used to analyse the data. Apart from that, we could see a significant decline after treatment with dupilumab. Therefore, it is reasonable to believe that serum levels of eotaxin-3 decreased from week 2 to week 52 after dupilumab treatment, regardless of the disease type (i.e., asthma or COVID-19). At the same time, we also noticed that the level of eotaxin-3 in patients with asthma before treatment was much higher than that in patients with AD and COVID-19, which indicated that eotaxin-3 was secreted more to recruit eosinophils to participate in the pathogenesis of asthma.
For PARC levels, only one study of AD was analysed, and as seen from the data, PARC levels decreased significantly after treatment with dupilumab at weeks 4, 8, 12 and 16. Since PARC could be induced by IL-4, IL-13 and IL-10, the inhibition of IL-4 and IL-13 led to a decrease in PARC serum levels, which further confirmed the interaction between IL-4/13 and PARC.
For the continuous variable-type meta-analysis, the data could be analysed only in the form of the mean ± SD. However, some of the studies did not display the data in this form directly due to the non-normal distribution of the raw data. Furthermore, some data were shown only in figures; therefore, we used the software GetData Graph Digitizer to capture the numerical values and convert them into other forms if needed. For example, in the study by Ariëns et al. [11], the median and interquartile range were demonstrated in the figure, so we used the GetData Graph Digitizer software to obtain data in the form of the mean ± SD.
Although the findings of our study were interesting, there are some limitations of our study. First, although many clinical studies have been published, little attention has been given to chemokines, which limited the performance of further analyses in our study. Second, only four studies involving different diseases were included, which made it difficult to further analyse the relationship between eotaxin-3 and PARC levels and disease severity. We also noticed that in our study, eotaxin-3 and PARC levels after dupilumab treatment were noted only in patients with classic type 2 inflammatory diseases; however, real-world effects of dupilumab on urticaria [19], bullous pemphigoid [20], and Netherton syndrome [21] have been observed. Although these studies were primarily case reports or case series, the administration of dupilumab is expanding, so more studies are needed to investigate changes in biomarkers for these diseases.