Summary
In the treatment of premature ventricular contraction (PVC), entirely treatment is achieved with radiofrequency ablation (RF). Not having enough PVC during three-dimensional mapping reduces the likelihood of ablation success. In our study, we showed that late potential mapping can be performed with fixed pacing from the right ventricle in PVCs originating from the right ventricular outflow tract. We observed that, in addition to ablation of the earliest point in local activation time mapping, ablation of the most delayed signals detected with constant pacing increases the chance of success in the long term.
Introduction
Premature ventricular contraction (PVC) is the most encountered form of idiopathic ventricular arrhythmias. Although generally known to be benign, it can cause malignant arrhythmias ranging from asymptomatic arrhythmias to severe heart failure [1].
Although radiofrequency ablation is generally preferred for the right ventricular outflow tract (RVOT) and fascicular PVCs if there is ≥ 20% PVC burden on 24-hour Holter recording, in some patients, the number of PVCs may show diurnal variation and radiofrequency ablation (RFA) may not be possible [2]. In these patients, either ablation is postponed, or a different drug treatment is considered.
It is known that isochronal late activation mapping (ILAM) in the sinus rhythm is used in RFA treatment of atrial and ventricular tachycardias documented but not induced during the electrophysiological study (EPS) [3–6]. In late potential mapping, it was observed that fragmented recordings were prolonged by constant pacing, and it was suggested that these areas may actively participate in tachycardia [7]. In addition, it has been said that late potential may be present in some of the limited number of outflow tract PVCs performed in the past [8, 9].
Mapping and RFA of PVCs that show diurnal changes during the day, and which are rare during 3D mapping has become very difficult [10, 11]. PASOTM, which is a method frequently used in PVC ablation in daily practice, based on the similarity with pacing at the point of origin or nearby locations, has some problems. Problems such as PVCs not always originating from a point where pacing can be performed or high amplitude and low similarity rates during pacing may be encountered.
In a previous study, it was reported that in patients with PVCs, the arrhythmogenic substrate may manifest itself electroanatomically in the sinus rhythm, either hidden in the QRS or slightly delayed at the end [12]. According to our hypothesis, fixed pacing from the right ventricular apex (RVA) may delay the separation of these fragmented signals from the QRS, and the point at which the most delayed signals are obtained may coincide with or be remarkably close to the earliest point during PVC. We call this method, which has not been used before in PVC mapping, Secret Signal Delayed Mapping (SSDM).
Aim
Our aim in this study is to determine the delay of fragmented signals with fixed pacing from the RVA during the sinus rhythm in patients with rare PVCs during 3D mapping using the method we called SSDM and to ablate the most delayed signal and compare it with the classical PVC ablation method.
Material and methods
Patient selection
In our prospective study, symptomatic patients with > 10% PVC detected in 24-hour rhythm Holter recordings and admitted to the electrophysiology laboratory for radiofrequency ablation between February 2021 and February 2023 in the arrhythmia clinic were included. R transition in PVC was after V3 in all patients. The patients in the first group underwent the earliest signal ablation and were called the local activation time (LAT) group. In the other group, both LAT and SSDM signal ablation was performed and it was named as the SSDM group. Patients with structural heart disease and genetic arrhythmic syndrome were excluded. The necessary permissions for the study were obtained from the ethics committee of the Adana City Training and Research Hospital.
Evaluation of laboratory findings
Renal function tests, total cholesterol, low-density cholesterol (LDL), high-density cholesterol (HDL), triglyceride, sodium, potassium, complete blood count, and thyroid stimulating hormone (TSH) levels were recorded.
Evaluation of 24-hour rhythm Holter monitoring
The 24-hour rhythm Holter recordings (SEER TM 1000 GE Medical Systems, Milwaukee, WI, USA) of all patients included in the study were analyzed before RFA. Inaccurate records redetected by the device in the rhythm Holter have been corrected. The total number of PVCs in 24 h was recorded.
Electrophysiological study, 3D mapping, induction, and radiofrequency ablation protocol
Patients with PVC > 10% on 24-hour rhythm Holter recordings were admitted to the electrophysiology laboratory for RFA. Antiarrhythmic drugs previously taken by the patients were discontinued before five half-lives. Through the standard femoral approach, 8 French sheaths for mapping catheter were inserted from the right side, and 6 French catheters for coronary sinus (CS) cannulation were inserted from the left side via venous access. The PVCs were then mapped with a 3D mapping system (CARTO 3, Biosense Webster, Diamond Bar, CA, USA). The earliest local electrical signals were marked (Figure 1). From these points, the THERMOCOOL SF irrigated catheter (Biosense Webster, Diamond Bar, CA, USA) with RFA catheter between 30 and 40 watts, and after the PVC disappeared, radiofrequency ablation was continued for an average of 3 min until the impedance value was < 110 ohms. Isoproterenol, and programmed stimulation maneuvers were tried again and RFA was considered successful if the PVC was not visible. If the PVC was not sufficient for mapping during the procedure, the following maneuvers were performed in sequence: 1) Programmed stimulation of the high right atrium, coronary sinus, and ventricle; 2) If the pacing maneuver did not work, intravenous infusion of isoproterenol (1–5 µg/min) was administered, and programmed stimulation was tried again.
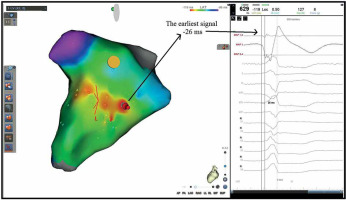
Figure 1
A patient with premature ventricular contraction (PVC) originating from the posterior right ventricular outflow tract, 18% PVC burden in 24-hour Holter monitoring. During local activation time (LAT) mapping, 42 points were collected in 91 min. LAT mapping measured -26 ms from the earliest point. Effective radiofrequency ablation was performed at the earliest signal and surrounding area in LAT mapping. Recurrence was observed in this patient after 5 months, and ablation was performed once more
Secret Signal Delayed Mapping (SSDM) technique in patients with premature ventricular contractions
After insertion of the mapping catheter, an average of 45–60 min was observed/it was observed for an average of 45–60 min and an average of 40–60 beats of PVCs were recorded during this period. A LAT map was created with these points. Then a 6F steerable Deca-polar CS catheter (Dynamic Deca, Bard Electrophysiology, Lowell, MA, USA) was placed in the right ventricular apex and fixed pacing was started (EP WorkMate Claris, St. Jude Medical, St. Paul, MI, USA). Device settings were adjusted to avoid interference between fixed pacing and PVC. Post-QRS delay was analyzed during pacing. An average of 128 ±24 points were included in the map. After the mapping process was completed, LAT and SSDM maps were compared (Figures 2, 3). The earliest point on the LAT map and the latest points on the SSDM map received adequate RFA. The procedure was concluded by carrying out the maneuvers that were mentioned earlier in the process.
Figure 2
A patient with premature ventricular contraction (PVC) originating from the anterior right ventricular outflow tract, 16% PVC burden in 24-hour Holter monitoring. During local activation time (LAT) mapping, 65 points were collected in 1 h. Then Secret Signal Delayed Mapping (SSDM) was started and 118 points were collected. A – LAT mapping measured -25 ms from the earliest point. B – The most delayed signal in SSDM mapping was measured as 76 ms. The distance between the earliest point in LAT mapping and the latest point in SSDM mapping was 5.1 mm. Effective radiofrequency ablation was performed at the earliest signal in LAT mapping, the most delayed signal in SSDM mapping, and between two points
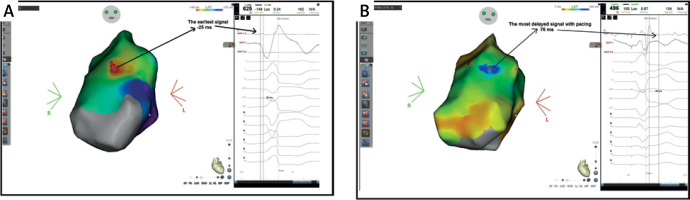
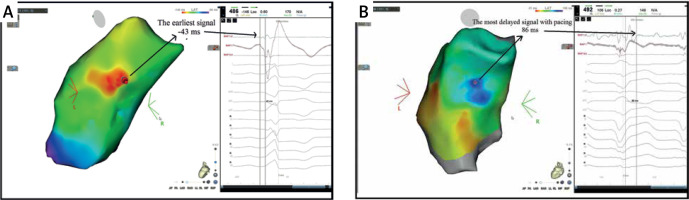
Figure 3
A patient with premature ventricular contraction (PVC) originating from the posterior right ventricular outflow tract, 19% PVC burden in 24-hour Holter monitoring. During local activation time (LAT) mapping, 101 points were collected in 1 h. Then Secret Signal Delayed Mapping (SSDM) mapping was started and 107 points were collected. A – LAT mapping measured -43 milliseconds from the earliest point. B – The most delayed signal in SSDM mapping was measured as 86 ms. The distance between the earliest point in LAT mapping and the latest point in SSDM mapping was 5.9 mm. Effective radiofrequency ablation was performed at the earliest signal in LAT mapping, the most delayed signal in SSDM mapping, and between two points
Clinical follow-up and identification of recurrences
Patients who underwent RFA were called for control in the first month and every 6 months thereafter. Patients with PVCs on ECG or persistent or discontinuous VT or > 1000 beats/day VPC on 24-hour rhythm Holter recordings were considered recurrent. Recurrent patients with > 10% VPC burden were recommended to undergo RFA again, while those with < 10% VPC burden were recommended to continue with drug therapy.
Statistical analysis
Data were analyzed using SPSS for Windows 20.0 (SPSS, Chicago, IL, USA). Variables were divided into categorical and continuous groups. Categorical variables were expressed as frequencies and percentages and were analyzed using the χ2 test. The Kolmogorov-Smirnov test was used to determine whether continuous variables had normal distribution or not. Continuous variables were expressed as mean ± standard deviation. Normally distributed variables were analyzed with independent samples t-test. Not normally distributed variables were analyzed with the Mann-Whitney U-test.
Results
We included 60 patients, 30 of whom underwent ablation according to the LAT and 30 according to the SSDM. In all patients, PVC originated from the RVOT. In the LAT group, 18 PVCs originated from the posteroseptal, 4 from the posterior, 5 from the anteroseptal, and 3 from the anterior RVOT. In the SSDM group, 17 originated from the posteroseptal, 6 from the posterior, and 7 from the anterior wall. The mean follow-up time of the patients was 10.2 ±1.6 months. Recurrence was seen in 11 (36.6%) patients in the LAT group and 4 (13.3%) patients in the SSDM group. Patient demographics, laboratory, echocardiographic, and 24-hour Holter monitoring findings and medication use were similar (Tables I–IV). When procedural findings were compared, total procedure time (p = 0.543) and total LAT points (p = 0.124) were similar in both groups. Signal earlyness in LAT mapping was significantly greater in the LAT group (p < 0.001) In the SSDM group, an average of 128 ±24 delayed signals were collected. The mean delayed signal time was 77.6 ±17.7 ms. In the SSDM group, the distance between the earliest signal on the LAT and the most delayed signal on the SSDM was 4.8 ±1.2 mm. In the SSDM group, the earliest and latest detected signals were not found at the same point in any patient.
Table I
Comparison of demographic and laboratory findings
Table II
Comparison of medications
Table III
Comparison of 24-hour rhythm Holter monitor findings
Discussion
In patients with PVCs originating from the RVOT, ablation is almost impossible if PVCs are not sufficiently observed during the procedure. In our study, it was shown that these PVCs may have signals secret in the ventricular electrogram (EGM). These signals can be separated from the ventricular EGM with a delay if there is fixed pacing from the RV. In addition to ablation of the earliest signals in the LAT mapping, it was found that lower recurrences were observed in patients who underwent ablation of the most delayed signals in the SSDM mapping.
In the study by Liu et al., it was reported that there may be hidden potentials in the sinus rhythm in outflow tract PVCs [12]. It was mentioned that these potentials were eliminated in most of the patients and recurrence rates were low. Although it has similar features to our study, all of the secret potentials were ablated in this study. In our opinion, PVC signals spread over the myocardium like other arrhythmias. Therefore, these signals can be detected in a large area. With our fixed pacing, we think that the center point of PVC can be detected by showing the separation and delay of the latent potentials from the QRS, and we think that it can be more successful with less ablation.
In another study, patients with outflow tract PVCs were compared with patients undergoing supraventricular tachycardia ablation as a control group [13]. It was reported that patients with PVCs may have local voltage recordings with a certain amplitude and width. In our study, local voltage recordings in the sinus rhythm were not investigated. As in the previous study, local voltage recordings could have been found in the region of delayed latent potentials.
In another study including right ventricular outflow tract PVCs, it was claimed that delayed potentials could also be detected in the sinus rhythm [8]. It was mentioned that arrhythmic substrates may be present in these regions and should be eliminated by ablation. This situation, which is mostly encountered in arrhythmias of ischemic origin, points to a slow conduction area. Therefore, we think that all of these slow-conduction areas cannot participate in PVCs, as in ischemic VTs. Ablation of bystander areas may create an arrhythmogenic substrate. With a fixed pacing maneuver, we think that the slowest conduction area in these areas and therefore the latest areas may be the site of origin of PVCs.
In another study, Liu et al. claimed that diastolic potential was observed at ideal ablation points in patients with PVCs originating from the RVOT [9]. They said that the resolution of the ablation catheter should be increased to visualize this signal. As known, diastolic potentials are obtained in the isthmus region during tachycardia in reentrant arrhythmias. Since PVCs are usually focal arrhythmias, we would not expect to see such a signal. We did not observe such signals in any of the patients in our study. Despite pacing, the latency of the latent potentials was measured to be 70–80 ms at most.
In another electroanatomic mapping study performed on patients with PVC, it was claimed that the points taken during PVC were displaced by an average of 6 mm according to the sinus rhythm [14]. In our study, a mean distance of 5 mm was found between the earliest point on LAT mapping and the latest point on SSDM mapping. The early point was taken during PVC and the late point was taken during pacing from the RVA. Although there seems to be a distance between them as claimed by the previous study, it is thought that it may be closer or originate from the same region.
There are studies suggesting that software systems (PaSo software, Carto 3, Biosense Webster, Diamond Bar, CA, USA), which look at the similarity rate with PVC by pacing with the ablation catheter, may help in determining the ideal ablation site and more successful ablation can be performed [14, 15]. As it is known, it is not possible to pace everywhere in the RVOT region. Therefore, it may not be possible to use this method in the region with weak signals. In addition, when high amplitude pacing is performed, similarity rates may decrease. In our method, we do not think that such problems can be encountered because of constant pacing from the RV.
Limitations: In our opinion, the biggest limitation is that to use the SSDM method, a small number of PVCs must be observed and correlated with the LAT map. If it is not correlated with the LAT map, false delayed signals due to pacing or other artifacts in very different regions may be marked. Our patient population was relatively small. During RV pacing, the patient may have palpitations and may be uncomfortable. The patients were not randomly assigned to different procedure groups.








