Introduction
Surgical site infection (SSI), as defined by the Centres for Disease Control and Prevention (CDC), is an infection that arises within 30 days of surgery or 1 year following implant placement [1–3]. Skin and subcutaneous tissue (superficial incisional), deep soft tissue (deep incisional), and other anatomical sites (organ/space) may be separated into several kinds of surgical site infections (SSIs) [4]. The most prevalent origins of SSIs [5] are bacterial colonisation of the skin, gastrointestinal system, and vaginal tract of patients. Compared to other kinds of surgery, abdominal procedures are frequently conducted with a clean-contaminated or contaminated incisional wound and are associated with SSI rates ranging from 15% to 25% [6–8]. SSI is one of the most frequent complications following abdominal surgery, and it substantially increases the risk of postoperative disability, fatalities, and financial burden [9–11]. To enhance patients’ quality of life and reduce their total medical expenditures, it is vital to reduce the frequency of their SSI claims. For more than 50 years, single- or double-ring wound edge protectors (WEPs) have been routinely used to shield the incisional wound and considered to lower the incidence of SSIs in surgical patients [12]. This is because cutaneous and gastrointestinal endogenous infections are the major causes of postoperative SSIs after abdominal surgery [13] and physical WEPs dramatically reduce the incidence of SSI by blocking sources of infection, such as intestinal material from entering the wound. Although wound edge protection solutions are highly successful in avoiding bacterial invasion, it remains unclear [14] whether or not they may lower the incidence of SSIs after abdominal surgery. Despite the anticipated clinical benefit of WEPs in lowering the SSI rate and the actual clinical benefit of WEPs, multiple randomized clinical studies (RCTs) [14–25] have yielded contradictory results. For example, RCTs such as [15–18, 20, 21, 23, 24] proved the significant clinical benefit of WEPs, while RCTs such as [14, 19, 22, 25] reported that WEPs did not provide effective protection against SSI after abdominal surgery. In addition, previous systematic reviews and meta-analyses [26–29] reported contradictory conclusions regarding the use of WEPs in reducing the incidence of SSIs in patients undergoing abdominal surgery, necessitating our current re-evaluation. This meta-analysis demonstrates the clinical efficacy of WEPs in preventing SSIs in patients undergoing various abdominal surgical procedures based on the credible evidence.
Aim
This study aimed to investigate the therapeutic effectiveness of a WEP in lowering the rates of SSIs that occurred following abdominal surgery.
Material and methods
The present meta-analysis was undertaken following the Preferred Reporting Items of Systematic Reviews and Meta-Analyses (PRISMA) guidelines [30].
Data sources and searches
An inclusive literature search was conducted without any restrictions on the year and language of publication utilizing the electronic databases Cochrane Library, EMBASE, and PubMed up to October 2022. In addition, relevant meta-analyses and studies’ bibliographies references were also searched. The search strategy involved the combination of the following key words: “Abdominal surgery”, “pancreatoduodenectomy”, “colorectal surgery”, “wound edge protectors”, “WEPs”, “double ring WEP device”, “single ring WEP device”, “post-operative surgical site infections”, “SSIs”, “Risk ratios”, “RRs”, “Meta-analysis”, “Randomized controlled trials”, “RCTs”. First, duplicate articles were deleted from the search results, followed by a title and abstract screening of the remaining articles. Finally, the full texts of all eligible studies were retrieved and reviewed for inclusion and exclusion based on the inclusion and exclusion criteria.
Study selection
The literature search was conducted separately by two authors. Through discussion, a consensus was obtained in the event of dispute. The following conditions must be met for a study to qualify: (a) randomised controlled trials examining the efficacy of WEPs vs. standard procedures with no use of WEPs and (b) studies evaluating the primary outcome of presence or absence of an SSI after the abdominal surgery. Exclusion criteria included randomised clinical trials that were carried out on healthy volunteers or those that involved patients who had had procedures other than abdominal ones, animal studies, reviews, letters, meeting reports, or comments. In addition, the following pre-specified subgroup analyses (effect of WEP versus non-WEP on the SSI rate) were performed: for different types of WEPs (single-ring and double-ring) and for different abdominal surgical sites (colorectal, upper digestive tract/small intestine, hepatobiliary and pancreatic, and appendix).
Data extraction
A computerised data extraction form was developed in Microsoft Excel and utilised for the purpose of documenting the fundamental information of the studies selected for the meta-analysis. This included the first author’s name, the year of publication, surgical site, sample size (intervention vs. control) and the conclusion. Two different authors independently extracted the data, and then the results of both authors’ extractions were compared. In the case of divergent opinions, an agreement was obtained via discussion. Depending on the circumstances, a third author was also included.
Quality assessment
The Cochrane Risk of Bias tool was applied in order to evaluate the methodological validity of each and every study that was incorporated into the meta-analysis. During the process of data extraction, selected articles were given a score, and RevMan version 5.4 [31] was used to construct a quality evaluation graph.
Statistical analysis
RevMan version 5.4.0 and MedCalc software [32] were used throughout the data processing procedure. The Mantel-Haenszel approach with the random effect model [33] was used in order to calculate the dichotomous pooled risk ratio and the 95% confidence interval (CI) for each outcome. A result was considered statistically significant if its p-value was less than 0.05 [34]. Forest plots [35] were used to visually represent the risk ratio and the 95% confidence range. I2 statistics [36] and χ2 test [37] were used to assess the level of heterogeneity present in the study’s results.
Results
Literature search results
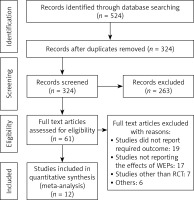
Figure 1 depicts the PRISMA chart for the selection of research. Through a comprehensive search of online databases, 524 studies were identified in total. After eliminating duplicates, the abstracts and titles of 324 studies were screened. Only 61 studies qualified for full-text evaluation. Twelve publications were finally included in the present meta-analysis based on the PICOS criteria [38] presented in Table I. Among the 12 studies, 5 studies were related to the post-operative SSIs after colorectal surgery with a total of 588 patients, 3 studies were related to pancreatoduodenectomy with a total of 6288 patients and 4 studies were related to the abdominal surgeries with a total of 2762 patients. These studies evaluated the effects of different ring types of WEPs with 7 studies using the single-ring type while 5 used the double-ring type of WEPs. The characteristics of all included trials including patients are displayed in Table II. Included studies evaluated the effectiveness of WEPs for reducing the post-operative SSIs in patients of abdominal surgery. In all included investigations, the presence or absence of an SSI after the abdominal surgery was measured as the primary outcome.
Table I
PICOS Search
Table II
General characteristics of studies in the final analysis
| No. | Study | Surgical sites | Ring type | Sample size (intervention versus control) | Conclusion (efficacy of WEPs)# | Reference |
|---|---|---|---|---|---|---|
| 1 | Baier, 2012 | Colorectal resection | Single | 199 (98 vs. 101) | None | [14] |
| 2 | Bressan, 2018 | Pancreatoduodenectomy | Double | 107 (57 vs. 50) | Yes | [15] |
| 3 | Cheng, 2012 | Colorectal resection | Double | 64 (34 vs. 30) | Yes | [16] |
| 4 | Imamura, 2016 | Abdominal surgeries | Single | 401 (199 vs. 202) | Yes | [17] |
| 5 | Kobayashi, 2018 | Colorectal surgery | Single | 102 (51 vs. 51) | Yes | [18] |
| 6 | Lauscher, 2012 | Colorectal resection | Single | 93 (46 vs. 47) | None | [19] |
| 7 | Liu, 2018 | Pancreatoduodenectomy | Double | 5969 (4756 vs. 1213) | Yes | [20] |
| 8 | Mihaljevic, 2015 | Abdominal surgeries | Single | 546 (274 vs. 272) | Yes | [21] |
| 9 | De Pastena, 2020 | Pancreatoduodenectomy | Double | 212 (94 vs. 96) | None | [22] |
| 10 | Pinkney, 2013 | Abdominal surgeries | Single | 735 (396 vs. 366) | None | [23] |
| 11 | Reid, 2010 | Colorectal surgery | Double | 130 (64 vs. 66) | Yes | [24] |
| 12 | Tsujinaka, 2013 | Abdominal surgeries | Single | 1080 (562 vs. 512) | None | [25] |
Risk of bias assessment and publication bias
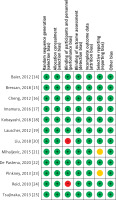
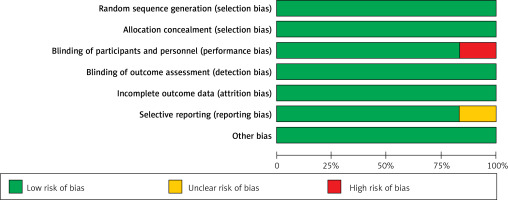
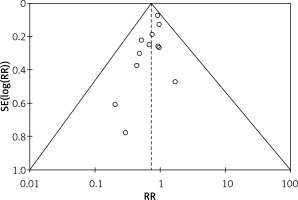
The quality assessment of the included studies was assessed as shown in Table III. Figure 2 depicts a summary of the risk of bias, whereas Figure 3 depicts a graph showing the risk of bias. Eight of the ten included studies had a low risk of bias whereas two studies had a moderate risk attributable to selective reporting or reporting bias and two studies posed a high risk of allocation concealment or selection bias. Figure 4 depicts the funnel plot, which indicated a low probability of publication bias with a significant p-value of 0.419 for Begg’s test [39].
Table III
Risk assessment for included studies
| Study ID and year | Baier, 2012 [14] | Bressan, 2018 [15] | Cheng, 2012 [16] | Imamura, 2016 [17] | Kobayashi, 2018 [18] | Lauscher, 2012 [19] | Liu, 2018 [20] | Mihaljevic, 2015 [21] | De Pastena, 2020 [22] | Pinkney, 2013 [23] | Reid, 2010 [24] | Tsujinaka, 2013 [25] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Was a consecutive or random sample of patients enrolled? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Did the study avoid inappropriate exclusions? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Did all patients receive the same reference standard? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Were all patients included in the analysis? | N | N | N | N | N | N | N | N | N | N | N | N |
| Was the sample frame appropriate to address the target population? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Were study participants sampled in an appropriate way? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Were the study subjects and the setting described in detail? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Were valid methods used for the identification of the condition? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Was the condition measured in a standard, reliable way for all participants? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
Efficacy outcomes
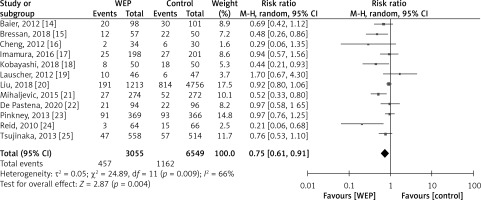
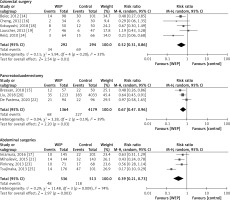
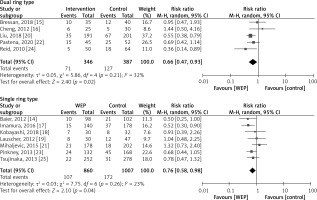
There was a total of 9,636 patients throughout all twelve trials that were conducted to give information on the clinical efficacy of WEPs in reducing the incidence of SSIs in patients undergoing abdominal surgery. Figure 5 displays a detailed pairwise comparison of each treatment group with respect to the main outcome: presence or absence of an SSI after the abdominal surgery. WEPs substantially decreased the post-operative SSIs with a risk ratio of RR = 0.75; 95% CI: 0.61–0.91; p = 0.004. Figure 6 demonstrates that WEPs are efficient in lowering the incidence of SSI at various abdominal surgical sites, with RR = 0.67; 95% CI: 0.47–0.96; p = 0.03 for pancreatoduodenectomy, RR = 0.52; 95% CI: 0.31–0.86; p = 0.01 for colorectal surgery, and RR = 0.39; 95% CI: 0.21–0.73; p = 0.003 for abdominal surgery. Furthermore, both kinds of WEPs (single-ring and double-ring devices) were effective in lowering the risk of SSIs, with RR = 0.66; 95% CI: 0.47–0.93; p = 0.02 for double-ring device and RR = 0.76; 95% CI: 0.58–0.98; p = 0.04 for single-ring device as shown in Figure 7. Risk ratio values less than 1 are prognostic of a high likelihood of WEPs in reducing post-operative SSIs in abdominal surgery patients. High heterogeneity was detected amongst pooled studies (I2 > 60%).
Discussion
This study was aimed to determine whether or not WEPs are effective therapeutically in avoiding SSIs in individuals who had abdominal surgery. Our comprehensive meta-analysis of 12 RCTs that included 9636 patients revealed that WEP is considerably beneficial in reducing the occurrence of SSI in patients who had undergone abdominal surgery.
The influence of WEP on reducing different degrees of SSI, as described by the CDC (superficial, deep, and organ/space), was investigated since it was shown that WEPs were more useful for superficial SSIs. After abdominal surgery, the number of superficial SSIs was significantly decreased by using WEP, however the effectiveness of WEP in reducing the number of deep and intrinsic SSIs remained equivocal. This might be due to the failure of WEP to protect the underlying tissue and the abdominal cavity from pathogenic sources, such as the leakage of gastrointestinal contents [40, 41].
Wound edges are the steep cliff-like epidermal edge that develops due to the lack of epithelial migration over a firm, pink, and level granulation base and responsible for delayed wound healing and a high risk of pathogen infiltration and septic shock [42]. WEPs/wound guards/wound retractors have been utilized for more than 50 years to seal these wound edges and lower the occurrence of SSI following abdominal surgery [43]. As a consequence, several WEP devices have been developed; nonetheless, they may be categorized into two broad groups: single- and double-ring ones.
Prior research has shown that double-ring devices are more effective in reducing the occurrence of SSI [44]; however, there are little data available on double-ring devices; hence, adequate rigorous high-quality testing is required. Based on the statistically significant findings of this meta-analysis, both single- and double-ring WEP significantly decreased the risk of SSI after abdominal surgery.
Colorectal surgery is often linked with higher SSI rates, ranging from 23 to 45%, as compared to any other surgery [45]. Borejsza-Wysocki et al. [46] reported in their article that closed incision negative pressure wound therapy have no benefit in reducing SSI after the surgical procedure while Shi et al. [47] mentioned that povidone-iodine has a beneficial role in decreasing the incidence of surgical site infections in the preoperative or intraoperative surgical procedures. Laparoscopy is a sort of minimally invasive surgery that does not require the surgeon to create significant incisions in the patient’s skin in order to view the internal structures of the patient’s abdomen (tummy) and pelvis. Its main advantage is that it avoids major incisions, which in turn reduces blood loss, pain, and discomfort. Less pain means less analgesia, which means fewer side effects for patients. Less damage and blood loss will occur due to the delicate nature of the equipment. Since these procedures use a minimally invasive approach [48] they are beneficial for reducing wound-related complications such as infection, dehiscence, and pain as they execute major surgeries through smaller incisions with compact, high-tech imaging systems than those required for traditional methods. This method was developed in the 1980s and has become increasingly popular in recent years. However, these surgical technologies are very expensive, require a great deal of technical skills, rely mostly on remote vision, experience a loss of haptic input, and primarily rely on hand-eye coordination. Therefore, the use of WEPs is still suggested. According to the subgroup analyses of several RCTs, WEPs are more effective in preventing SSIs in clean-contaminated and colorectal operations [49, 50]. The vast majority of colorectal procedures are clean-contaminated cases, which were by far the most common kind of surgery performed in the included RCTs. The results of this meta-analysis are comparable to those of previously published systematic reviews [51, 52] and concluded that wound protectors significantly decreased the incidence of surgical site infections following abdominal surgery.
Limitation: There are certain limitations to this meta-analysis. First, while this analysis was conducted with the suggested methodological rigour, the results are constrained by the availability of just 12 RCTs with moderate to high heterogeneity. Second, risk ratio values were mostly employed to determine the association between the two medications, which may introduce bias when comparing the outcomes of randomised, controlled trials of varying durations. Finally, in terms of manufacturing materials, shapes or forms, and specific functions, it was not possible to unify WEPs. Similarly, the multifactorial process in which SSI occurs, altering a single factor (wound protection) may not provide significant results. Increased use of WEPs over time may have contributed to differences in results among studies conducted at various times. Therefore, there is a huge need for a more logical design and rigorous execution of large-scale multicentre RCTs, especially with regard to WEP’s effectiveness in avoiding SSI in patients having abdominal surgery with diverse abdominal surgical locations, varied surgical modalities, and varying contamination levels.
Conclusions
Based on the statistically significant findings, this meta-analysis concludes that both double- or single-ring wound edge protection devices are successful in preventing surgical site infections following pancreatoduodenectomy, colorectal, and abdominal surgery. These devices are safe and succinct interventions that may help reduce postoperative morbidity and mortality.