A 66-year-old woman was referred to our hospital for valve-in-valve (ViV) transcatheter aortic valve implantation (TAVI). She had a history of severe bicuspid aortic valve stenosis, undergoing surgical replacement with a bioprosthesis in 2015. Two years after that she underwent REDO surgery to replace the aortic valve and ascending aorta with Bentall aortic homograft no. 23 for prosthesis endocarditis. The patient was asymptomatic until March 2021 when echocardiography showed severe stenosis for valve deterioration. At pre-procedural computed tomography (CT) the patient was found to be at high risk of left coronary artery occlusion (CAO) [1–3] (Supplementary Figure S1 A–C), so we opted for a transfemoral TAVI using a self-expandable valve with coronary artery protection. The left main (LM) was cannulated with an EBU catheter 3.5 from the left femoral artery and a Xience stent 4.0 × 18 mm was put in the mid left anterior descending artery (LAD). A CoreValve Evolut Pro+ 26 mm was introduced and, after correct positioning, was partially opened during ventricular pacing at 120 bpm. Since angiography did not indicate risk of CAO, we slowly retracted the EBU catheter without removing the LAD wire and completed valve deployment. Angiographic control showed an image at risk of left CAO, so we performed modified chimney stenting – the “tunnel technique”. We re-accessed the LM with another EBU catheter, leaving the wire in the LAD, then, after LAD rewiring, a Xience 4.0 × 12 mm stent and an Onyx 4.5 × 15 mm stent were deployed in overlap from the LM ostium to the Core-Valve frame, creating a protective tunnel. All the phases of tunnel stent technique are shown in Figures 1 and 2, where they are compared with chimney stenting ones. At post-procedural CT also with three-dimensional reconstruction a significant protrusion of the tunnel stent in the aortic lumen was excluded (Supplementary Figure S1 D).
Figure 1
The tunnel stent technique starts as a chimney stenting procedure with left coronary artery cannulation, coronary wiring and stent passage through the wire in the mid LAD after which the aortic prosthesis is positioned in cusp overlap projection (LAO 19° and caudal 16°) (A); opening of about 2/3 of the prosthesis during ventricular pacing at 120 bpm in left oblique projection (LAO 36°) with coronary angiographic control and release of the valve in cusp overlap (B); final angiography showing an image at risk of CAO. After the retraction of the first catheter, leaving the coronary wire in situ, left coronary is re‑accessed with another catheter through the cells of the prosthetic valve frame and another wire is put in the coronary (C). One or more stents are implanted between the frame of the aortic prosthesis and the left coronary ostium (D)
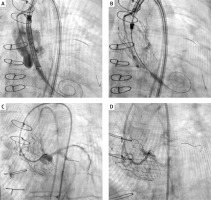
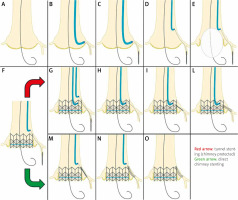
Figure 2
Panels A to F present the common steps of the tunnel technique and chimney stenting with the red arrow indicating from panels G to L the final steps of tunnel stenting (chimney protected) as described in Figure 1 legend; the green arrow indicates instead the final steps of direct chimney stenting with stent retraction (M) as well as stent dilation and final release (N, O)
With this case we present an alternative strategy to chimney stenting and to BASILICA (bioprosthetic or native aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction) technique [4] for ViV TAVI at high risk of CAO. Tunnel technique is more complex than chimney technique but has several advantages (Table I). Its aim is to create a continuous tunnel between the coronary ostia and the prosthesis frame, which should be as straight as possible and, even if some curves are present in its course, these are straighter than the angle created with the chimney technique, possibly leading to a reduction in the incidence of stent thrombosis [5, 6].
Table I
Advantages and disadvantages of tunnel vs. chimney stenting technique
However, this procedure also has several downsides (Table I). First, when retrieving the catheter from the LM, though leaving the coronary wire in situ, before re-accessing the coronary, acute CAO may occur, so in patients at higher risk of CAO the chimney technique should be preferred.
Second, the tunnel technique is longer than the chimney technique and may require a larger amount of contrast medium, so it should not be chosen in patients at higher risk of contrast-induced nephropathy.
Finally, coronary re-access may be difficult and an optimal alignment with the coronary ostium may not always be possible: this should not lead to making multiple attempts of coronary re-access to avoid complications.
For all these reasons, the choice of this technique requires a careful selection of patients who could benefit from it without intra- and peri-procedural complications.








