Introduction
Colorectal cancer (CRC) poses a significant threat to the life and health of Chinese residents, with morbidity and mortality ranking third and fifth, respectively, among all malignant tumors [1]. Over the past decade, China has witnessed an increasing burden of colorectal cancer, prompting national and local governments to promote public health service projects and carry out population screening initiatives in more areas. As a result, early diagnosis rates have been on the rise. Studies indicate that treatment techniques such as endoscopic mucosal dissection and mucosal resection have proven to be effective in treating early gastroenteric tumors, with therapeutic outcomes comparable to those of surgery [2, 3]. However, as the indications for endoscopic therapy continue to expand and the incidence of non-complete curative resection of early colorectal cancer increases, additional treatments are needed to effectively control the risk of local recurrence and lymph node metastasis of cancer.
A standard method for performing additional surgery after endoscopic submucosal dissection (ESD) for early colorectal cancer has yet to be established. In today’s era of functional protection and precision treatment, we advocate for more minimally invasive and individualized treatment options, including reduced lymph node dissection and reduced-port laparoscopic surgery. Some authors have compared the clinical application of conventional laparoscopic surgery with natural orifice specimen extraction surgery (NOSES) and single-incision plus one-port laparoscopic surgery in colorectal cancer to demonstrate the oncological equivalence of these methods [4–6].
Aim
To enhance the understanding of different surgical outcomes by discussing additional treatment strategies following non-complete curative endoscopic resection of early colorectal cancer. We compared the treatment outcomes of conventional laparoscopic surgery (CLS) with various reduced-port laparoscopic surgical procedures, including single-incision plus one-port laparoscopic surgery (SILS+1) and three-port laparoscopic surgery combined with natural orifice specimen extraction surgery (three-port NOSES). We aimed to explore the short-term outcomes, cosmetic effects, and social effects of these two types of reduced-port laparoscopic surgery.
Material and methods
From January 2018 to December 2022, a total of 88 patients who had undergone non-curative endoscopic resection of early colorectal cancer received additional surgical procedures. The indications for additional surgery were as follows: (1) resection specimens with positive lateral or basal margins, (2) high infiltration of the submucosa (more than 1000 µm), (3) lymphovascular infiltration, (4) poor differentiation adenocarcinoma, undifferentiated carcinoma, or mucus adenocarcinoma, (5) carcinoma sprouting grade of G2 or more, (6) incomplete resection margin evaluation or fragmented specimens, and (7) inconclusive pathological results with a decision to undergo surgery made in consultation with the patient. We excluded patients who underwent emergency surgery for acute perforation or bleeding. The study was conducted in accordance with the Declaration of Helsinki. All patients were informed of the risks associated with additional surgery, and both the patients and their family members signed informed consent forms. The retrospective study was approved by our hospital’s Ethical Review Board.
The patients were divided into three groups based on the surgical method: conventional laparoscopic surgery, single-incision plus one-port laparoscopic surgery (Photo 1), and three-port laparoscopic surgery combined with natural orifice specimen extraction surgery (Photo 2).
Photo 1
Single-incision plus one-port laparoscopic surgery. A – Operative photograph of single-incision plus one-port laparoscopic surgery (SILS+1). B – Figure of abdominal wall incision after single-incision plus one-port laparoscopic surgery (SILS+1)
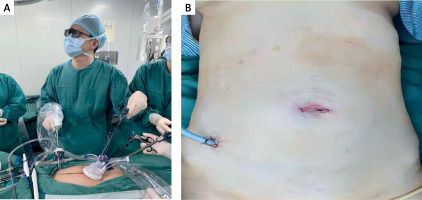
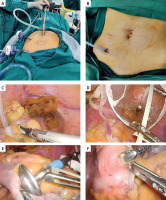
Photo 2
Three-port laparoscopic surgery combined with natural orifice specimen extraction surgery (threeport NOSES). A – Operative photograph of three-port NOSES. B – Figure of abdominal wall incision after three-port NOSES. C – Ligation and closure of the distal colon with plastic strap in three-port NOSES. D – Pulling the surgical specimen and plastic strap out of the body through the anus. E – Inserting a stapler with thread into the proximal colon. F – After intestinal wall incision, the stapler is pulled out of the intestinal canal through thread traction
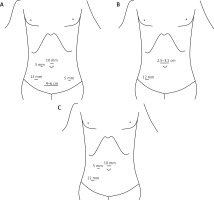
The placement of the surgical incisions are as follows (Figure 1): 1. For the CLS procedure: the observation port is positioned 1 cm above the navel (10 mm). The main operating port is situated 3 cm inward from the right anterior superior iliac spine (12 mm). Secondary operating ports are positioned on the midline between the right or left clavicle and the navel (5 mm), and 3 cm inward from the left anterior superior iliac spine (5 mm). The specimen extraction incision is positioned 3 cm above the pubic symphysis, with a horizontal incision extending 4–6 cm. 2. For the SILS+1 procedure: a single-port trocar is positioned above the upper edge of the navel (2.5–3.5 cm). The main operating port is located 3 cm inward from the right anterior superior iliac spine (12 mm). 3. For the three-port NOSES procedure: the observation port is positioned 1 cm above the navel (10 mm). The main operating port is located 3 cm inward from the right anterior superior iliac spine (12 mm). The secondary operating port is positioned on the midline between the right clavicle and the navel (5 mm).
Figure 1
Placement of the surgical incisions. A – Conventional laparoscopic surgery (CLS). B – Single-incision plus one-port laparoscopic surgery (SILS+1). C – Three-port laparoscopic surgery combined with natural orifice specimen extraction surgery (three-port NOSES)
The preoperative patient characteristics, including age, gender, body mass index (BMI), American Society of Anesthesiologists (ASA) score, and tumor location were recorded. Postoperative outcomes included operation time, blood loss, first exhaust and diet time, length of abdominal incisions, complications, length of hospital stay, and pathological results. The Visual Analog Scale (VAS) is utilized to quantify patients’ pain perception, with a score of 0 indicating no pain and a score of 10 representing the most severe pain. Patient satisfaction with the incision is used as a measure of cosmetic result, with a maximum score of 10 indicating complete satisfaction. Preoperative endoscopic nanocarbon localization effectively marked the original location of the lesion and provided precise guidance for subsequent additional surgery in terms of lesion localization.
Statistical analysis
The statistical analysis was performed using SPSS 26.0 software. Continuous data with a normal distribution were described as mean ± standard deviation (SD). To compare the differences, a one-way ANOVA was applied. For data that did not follow a normal distribution, median [interquartile range] was used, and the differences were assessed using the Mann-Whitney U test. Categorical data were presented as the number of cases (%) and differences were analyzed using the Χ2 test. A significance level of < 0.05 (two-sided) was considered statistically significant.
Results
This study included a total of 88 patients who required additional surgical treatment following non-curative endoscopic surgery for early colorectal cancer. Among these patients, 38 underwent CLS, 32 underwent SILS+1, and 18 underwent three-port NOSES. The baseline characteristics of the patients are presented in Table I. No significant differences were observed among the 3 groups in terms of sex, age, BMI, tumor location, or ASA class. The reasons for requiring additional surgery after ESD are summarized in Table II. Histological complete resection was achieved in 70 out of the 88 treated tumors. However, 10 (11.4%) specimens had positive margins, and 8 (9.1%) specimens were incompletely resected or had unevaluable margins. Among the cases, 2 (2.3%) had muscularis mucosae infiltration, 59 (67.0%) exhibited deep submucosal invasion (≥ 1000 µm), 6 (6.8%) showed invasive lymphovascular infiltration, and 3 (3.4%) were poorly differentiated cancers. All patients successfully underwent laparoscopic radical colorectal cancer surgery following preoperative nanocarbon endoscopy localization.
Table I
Baseline characteristics of patients
[i] Data are presented as mean ± standard deviation. CLS – conventional laparoscopic surgery, SILS+1 – single-incision plus one-port laparoscopic surgery, three-port NOSES – three-port laparoscopic surgery combined with natural orifice specimen extraction surgery, BMI – body mass index, ASA – American Society of Anesthesiologists.
Table II
Reasons for additional surgery after endoscopic submucosal dissection
Table III provides a summary of the intraoperative and postoperative outcomes. The analysis revealed significant differences in operation time among the three groups. Specifically, the CLS and SILS+1 groups exhibited significantly shorter operation times compared to the three-port NOSES group (114.500 [106.8,118.0], 110.000 [105.0,115.0], versus 124.500 [120.8,129.8]; p < 0.001). However, no significant differences were observed among the three groups in terms of blood loss, first postoperative exhaust or diet time, number of lymph nodes dissected, length of intestine resected, or complications. The SILS+1 and three-port NOSES groups exhibited significantly better postoperative outcomes in terms of cosmetic effect (4.000 [3.8,5.0], 7.000 [7.0,8.0] versus 7.000 [7.0,8.0]; p < 0.001), VAS score (4.000 [3.0,5.0], 3.500 [3.0,4.0] versus 3.000 [3.0,4.0]; p = 0.003) and postoperative hospital stay (8.000 [7.0,9.0], 7.000 [6.3,8.0.], versus 7.000 [6.3,8.0]; p = 0.035). Additionally, the three-port NOSES group had a statistically significant shorter abdominal incision length than the CLS and SILS+1 groups (7.11 ±0.38, 4.24 ±0.33, 3.16 ±0.22, p = 0.001).
Table III
Outcomes of additional surgery after non-curative ESD
| Variables | CLS (n = 38) | SILS+1 (n = 32) | Three-port NOSES (n = 18) | P-value |
|---|---|---|---|---|
| Operation time [min] | 114.5 [106.8,118.0] | 110.0 [105.0,115.0] | 124.5 [120.8,129.8]*** | < 0.001 |
| Blood loss [ml] | 15.0 [10.0,20.0] | 20.0 [10.0,20.0] | 17.5 [15.0,20.0] | 0.113 |
| First postoperative exhaust time [day] | 2.0 [1.5,2.5] | 2.0 [1.0,3.0] | 2.0 [2.0,3.0] | 0.097 |
| First postoperative diet time [day] | 2.0 [2.0,3.0] | 2.0 [2.0,2.0] | 2.0 [2.0,3.0] | 0.215 |
| Cosmetic scores | 4.000 [3.8,5.0]* | 7.000 [7.0,8.0] | 7.000 [7.0,8.0] | < 0.001 |
| VAS scores | 4.000 [3.0,5.0]* | 3.500 [3.0,4.0] | 3.000 [3.0,4.0] | 0.003 |
| Lymph node dissection | 16.05 ±6.97 | 15.88 ±6.57 | 17.50 ±6.72 | 0.692 |
| Lymph node dissection ≥ 12 | 24 (63.2%) | 23 (71.9%) | 14 (77.8%) | 0.501 |
| Length of abdominal incision [cm] | 7.11 ±0.38* | 4.24 ±0.33** | 3.16 ±0.22 | < 0.001 |
| Length of intestine resected [cm] | 15.47 ±2.02 | 15.44 ±2.02 | 16.44 ±1.54 | 0.160 |
| Length of hospital stay [days] | 8.000 [7.0,9.0]* | 7.000 [6.3,8.0.] | 7.000 [6.3,8.0] | 0.035 |
| Complication: | 4 (10.5%) | 3 (9.4%) | 2 (11.1%) | 0.983 |
| Anastomotic leakage | 1 | 1 | 0 | |
| Anastomotic bleeding | 0 | 0 | 1 | |
| Lymphatic leakage | 1 | 0 | 1 | |
| Intestinal obstruction | 1 | 1 | 0 | |
| Incision infection | 1 | 1 | 0 |
* The value of the CLS group was statistically significantly different compared with SILS+1 and three-port NOSES.
** The value of the SILS+1 group was statistically significantly different compared with three-port NOSES group.
*** The value of the three-port NOSES group was statistically significantly different compared with CLS and SILS+1 groups. CLS – conventional laparoscopic surgery, SILS+1 – single-incision plus one-port laparoscopic surgery, three-port NOSES – three-port laparoscopic surgery combined with natural orifice specimen extraction surgery, VAS – Visual Analog Scale.
Postoperative anastomotic leakage was reported in 2 patients (1 case each in the CLS and SILS+1 groups), and an additional transverse colostomy was performed in the SILS+1 group. Additionally, there was 1 case of anastomotic bleeding (three-port NOSES group), 2 cases of lymphatic leakage (1 case each in the CLS and three-port NOSES groups), 2 cases of intestinal obstruction and incision infection (1 case each in the SILS+1 and CLS groups), and 2 cases of incision infection (1 case each in the SILS+1 and CLS groups). There was no significant difference in the incidence of total complications between the groups. Postoperative pathological examination revealed local lymphatic node metastasis (LNM), nerve invasion, or intravascular cancer thrombus in 15 (17.0%) cases. The overall median follow-up period was 33 months, ranging from 5 to 56 months. During the follow-up period, 2 cases of liver metastasis were identified, and unfortunately, 1 patient died due to cerebral hemorrhage 1.5 years after the operation.
Discussion
The National Comprehensive Cancer Network (NCCN) guidelines recommend follow-up for T1 stage colorectal cancer after the complete endoscopic resection of a single whole specimen. The guidelines emphasize that factors such as submucosal invasion ≥ 1000 µm, lymphatic vascular infiltration, poor differentiation, tumor budding, and incomplete resection are independently associated with an increased risk of lymph node metastasis and residual cancer. Therefore, radical resection is considered necessary [7]. However, the optimal surgical approach for additional surgery after non-curative endoscopic resection for early colorectal cancer has not been clearly defined and remains controversial. The controversy mainly stems from the understanding of lymphatic drainage in early colorectal cancer and the question of whether the trauma caused by conventional laparoscopic radical resection for colorectal cancer is inevitable.
According to the Chinese Protocol of Diagnosis and Treatment of Colorectal Cancer (2015 edition), local resection is recommended for T1N0M0 stage tumors with favorable histological characteristics, such as good differentiation and no vascular infiltration [8]. However, the Japanese Classification of Colorectal Carcinoma suggests slightly different recommendations. For Tis (tumor in situ) in the clinical stage, local resection (D0) or segmental resection (D1) is feasible. D2 surgery is feasible for SM (T1) N0 stage, and D2 or D3 surgery is feasible for MP (T2) N0 stage [9]. Some scholars have studied the metastasis of the third station lymph node in different stages of colorectal cancer. It has been found that the third station lymph node in T1 stage does not metastasize, while the metastasis rates for T2, T3, and T4 stage tumors are 3.2%, 4.8%, and 8.9% respectively. This suggests that D2 radical resection is feasible for T1 stage left colon cancer, while D3 radical resection should be considered for T2-4 stage tumors [10]. There is also controversy regarding the extent of segmental resection for left colon cancer, specifically the length of intestinal canal resection at both ends of the tumor. According to the requirements of the 7th edition of the Japanese Classification of Colorectal Carcinoma, the cutting line should be determined based on the scope of intraoperative trophoblast vessels and lymph node dissection. Additionally, the scope of bowel segmental resection is determined by combining the principle of “10+5” [9]. Recent systematic reviews suggest that the metastasis rate of lymph nodes surrounding the intestinal canal, with a length of more than 10 cm, is only 1–2%. The range of lymph node metastasis around the distal colon and rectum rarely exceeds 5 cm [11]. Based on the above studies, we conducted a thorough literature search to investigate whether reduced-port laparoscopic surgery, in combination with colon segmental resection and D1 or D2 lymphatic dissection, is more beneficial in reducing surgical trauma and accelerating postoperative recovery. However, we failed to find corresponding studies addressing this specific topic.
In our study, we conducted an investigation into the oncology outcomes of 88 patients who were divided into three groups. These patients had undergone additional surgery after non-curative endoscopic resection for early colorectal cancer. Our main focus was to compare the outcomes of different surgical approaches, namely conventional laparoscopic surgery, single-incision plus one-port, and three-port NOSES. Our objective was to determine the most effective and minimally invasive surgical method for the radical treatment of colorectal cancer.
The minimum number of lymph nodes to be resected in standard radical colorectal cancer specimens is at least 12 for accurate postoperative staging and prognosis determination [12, 13]. Our study examined the number of lymph nodes resected among three groups and found no significant difference (16.05 ±6.97, 15.88 ±6.57, versus 17.50 ±6.72; p = 0.692; Table III). Additionally, there was no significant difference in the proportion of patients with more than 12 lymph nodes (63.2%, 71.9%, versus 77.8%; p = 0.501, Table III). Tao Chen [14] reported that non-curative ESD followed by additional surgery for early colorectal cancer was a safe approach with a 5.9% incidence of postoperative complications. Comparing the risk of lymph node metastasis with the incidence of complications, patients benefited from additional surgery after non-curative ESD. In our study, we found no significant difference in the complication rates among the three techniques (10.5%, 9.4%, versus 11.1%; p = 0.983, Table III). Therefore, no technique demonstrated superior safety in terms of complication rates or reoperation rates.
Based on the postoperative pathological results, it was confirmed that 17.0% of the patients requiring additional surgery had positive findings, including lymph node metastasis, tumor residue, and nerve vessel infiltration. These findings strongly support the necessity of performing the surgery. Cheng P’s study yielded similar findings, indicating that after ESD followed by additional surgery, 19.35% of patients were found to have residual cancer and lymph node metastasis [15]. In our study, 89.8% of the patients were in stage I after the operation. Among a total of 7 stage III patients with lymph node metastasis, 6 patients had D1 lymph node metastasis, and only 1 patient had D2 lymph node metastasis. Based on the data obtained, we propose the utilization of reduced-port laparoscopic surgery with minimized trauma for performing additional surgery after non-curative ESD. In this approach, the extent of lymph node dissection can be appropriately reduced, including the possibility of D2 or even D1 lymph node dissection. Both of the reduced-port laparoscopic techniques employed in our study are capable of effectively achieving resection of the affected bowel, regional lymph node dissection, and reconstruction of the digestive tract. As a result, our findings provide a solid basis for selecting optimal surgical approaches for managing early colorectal cancer cases that have undergone non-curative ESD.
In recent years, there has been growing recognition of the advantages of SILS+1 and three-port NOSES over CLS in terms of minimally invasive techniques and accelerated postoperative recovery [16, 17]. Multiple studies have investigated the short-term and long-term safety of reduced-port surgery compared to conventional laparoscopy in colorectal surgery. The findings consistently demonstrated that reduced-port surgery achieves similar pathological and long-term oncological outcomes compared to conventional laparoscopy [4, 18]. In our study, we conducted a comparison of the short-term surgical outcomes between the two types of reduced-port surgery and conventional laparoscopic surgery. The results showed no significant differences in blood loss, gastrointestinal functional recovery, or complications among the three groups. It has been reported that the NOSES technique provides benefits for patients with colorectal cancer, including faster recovery, shorter postoperative hospital stay, less incisional pain, and shorter scars [19]. Ju Myung Song reported that reduced port laparoscopic surgery (RPLS) demonstrated significant superiority over CLS in terms of postoperative pain [4]. In our study, we utilized VAS scores to evaluate postoperative pain and found that the reduced-port groups had lower postoperative pain scores compared to the conventional laparoscopic groups (4.000 [3.0,5.0], 3.500 [3.0,4.0] versus 3.000 [3.0,4.0]; p = 0.003). Additionally, the incision length in the reduced-port groups was significantly shorter than that in the CLS group (7.11 ±0.38, 4.24 ±0.33, 3.16± 0.22, p < 0.001), with the three-port NOSES group having the shortest incisions and the lowest level of pain. These findings suggest that, compared to traditional laparoscopic surgery, reduced-port surgery demonstrates milder incisional pain, higher incision satisfaction, shorter hospital stay, and ultimately better surgical outcomes.
To the best of our knowledge, this is the first retrospective cohort study that compares three distinct surgical approaches for the management of patients with early colorectal cancer following non-curative endoscopic resection. Although this study was conducted retrospectively at a single center, it provides evidence supporting the oncological viability of two reduced-port surgical techniques in comparison to conventional laparoscopic surgery. Furthermore, the reduced-port procedures demonstrated superior postoperative recovery and enhanced cosmetic outcomes. The main limitations of this study include its retrospective design and a relatively small number of clinical cases. Therefore, future research should focus on conducting multicenter prospective randomized controlled trials. At the same time, Zhang J’s study points out that transanal NOSES surgery can impair anal function [19], so indicators such as anal function should be included in further research.
Conclusions
Different types of reduced-port laparoscopic surgical strategies are effective and safe when applied to additional surgery for early colorectal cancer after non-curative endoscopic resection. With less postoperative pain, faster recovery, better cosmetic outcome, and higher patient satisfaction, which results in improved cost and social effectiveness, reduced-port laparoscopic surgery is expected to become the first choice treatment plan for additional surgery after non-curative endoscopic resection for early colorectal cancer.








