Diaphragmatic hernias can be described as translocations of abdominal organs or omental fat to the thoracic cavity through a diaphragm defect [1]. Depending on the cause, they can be divided into congenital and acquired (usually post-traumatic). Congenital hernias are typically found in neonates and infants. They approximately occur with the frequency of 2.5 cases per 10 000 births [2]. Anterior diaphragmatic hernias (Morgagni hernias) account for only 2–4% of them (Figure 1) [1]. The presence of such abnormalities might produce pulmonary, gastrointestinal and cardiac symptoms but it can also be asymptomatic. Here we present a case report of a patient who had a diaphragmatic hernia incidentally found during the coronary artery bypass graft surgery.
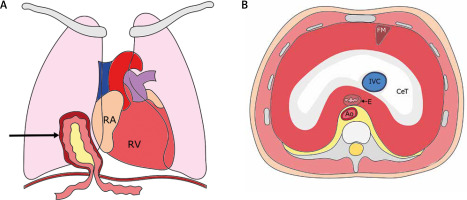
Figure 1
A – Frontal section of the thorax; black arrow indicates the hernia sac with the transverse colon and omentum inside. B – Transverse section of the thorax – superior view of the diaphragm
RA – right atrium, RV – right ventricle, FM – foramen of Morgagni, CeT – central tendon, IVC – inferior vena cava, E – esophagus, Ao – aorta.
A 64-year-old man presented with ventricular tachycardia was admitted to the hospital and successfully treated with electrical cardioversion. He underwent emergency coronary angiography that revealed chronic occlusion of the right coronary artery and circumflex branch of the left coronary artery, subtotal occlusion of the left anterior descending (LAD) artery and significant stenosis of the intermediate (IM) branch of the left coronary artery. The echocardiogram showed moderate impairment of left ventricle contractility with ejection fraction 39%. His cardiovascular and operative risk factors were type 2 diabetes, atherosclerotic peripheral arterial disease of the lower extremities, pulmonary emphysema (a long-time cigarettes smoker) and arterial hypertension. The patient underwent emergency, off-pump, beating heart bypass surgery through a median sternotomy. The right internal thoracic artery (RIMA) was anastomosed to the LAD and the left internal thoracic artery to the IM. The RIMA harvesting technique and the beating heart off-pump coronary grafting performed on the lateral wall of the heart required opening of the right pleural cavity. A right anterior diaphragmatic hernia was incidentally found during the operation. Before surgery, the patient did not report any complaints which could suggest the presence of the hernia and only the chest X-ray showed a shadowing in the right cardiophrenic angle (Figure 2). The sternotomy wound did not require extension inferiorly through the linea alba as it was described in previous case reports of larger diaphragmatic defects [3]. The hernia neck was incised, adhesions within the sac were divided to allow the contents (transverse colon with omentum) to be reduced and the sac was excised (Figure 3). The diaphragmatic defect was closed with interrupted 2-0 nylon sutures (Figure 4). The patient recovered uneventfully and was discharged home from the hospital.
Figure 2
Chest X-ray comparing the preoperative shadowing in the right cardiophrenic angle (A) and the improvement postoperatively (B)
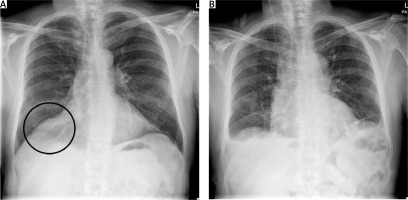
Figure 3
Intraoperative picture showing the anterior diaphragmatic defect responsible for the hernia (black arrow) and the excised hernia sac (white arrow)
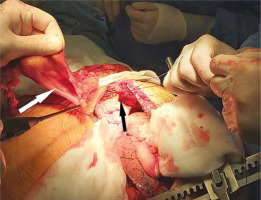
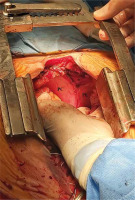
Figure 4
Intraoperative picture showing the sternocostal triangle (black star) after surgical repair of the diaphragmatic defect
PC – pericardium, PL – right pleura.
Case reports of diaphragmatic hernia in adult cardiac surgery patients have rarely been described. Brookes et al. presented a case of a male patient who was referred for coronary artery bypass graft surgery. A Morgagni hernia was revealed during preoperative diagnostic process. Because of the lack of symptoms the decision was made not to repair the hernia simultaneously. However, during surgery, the contents of the hernia became tense and there was a risk of their strangulation, so it was decided to undertake concomitant hernia repair [3]. Rose et al. also presented a finding of a diaphragmatic hernia, which was related to the patient’s heart surgery. Yet, in this article there is no description of the patient’s history and management of the hernia [4]. Another case report presented a woman with a post-traumatic intrapericardial hernia. The patient underwent surgery because of aortic aneurysm and a simultaneous repair of the hernia was also performed [5].
The majority of patients with Morgagni hernias are asymptomatic or their symptoms are not specific. They often complain of dyspnoea and other pulmonary symptoms, abdominal pain, nausea and vomiting. In some cases, a patient’s signs and symptoms can be suggestive of a cardiac tamponade. The diagnostic algorithm mostly involves an X-ray or computed tomography (CT) imaging [1, 6, 7]. Those hernias might be easily misdiagnosed or overlooked, especially when a patient presents with other apparent conditions requiring emergency management. In our case report, preoperative chest X-ray indicated a possibility of existence of a pathological mass in the right cardiophrenic angle, but a further diagnostic process was abandoned due to the unstable angina of the patient. Massive pulmonary emphysema could make the interpretation of the X-ray more difficult. However, a shadowing in the right cardiophrenic angle was noticed. In patients with diaphragmatic hernias, echocardiography might be complicated due to the presence of the organs filled with air in the thoracic cavity [8].
Definitive treatment of diaphragmatic hernias is surgical. Laparotomy, laparoscopy and thoracoscopy are possible approaches for repair operations. Transsternal access is indicated for patients who undergo concomitant cardiac surgery or when pericardial adhesions are present [7, 8]. It was used in our case. The diaphragmatic defect can be repaired using a mesh or it can be simply sutured [1, 5]. In procedures performed through a sternotomy it is possible to use a double-lumen endotracheal tube to maintain one-lung ventilation for a better surgical field [9].
Patients with diaphragmatic hernias who undergo cardiac surgery are prone to additional complications related to their condition. When the hernia is located directly posterior to the sternum, there is a risk of its damage during the sternotomy. It could result in release of the intestine contents to the thoracic cavity [10]. Another danger is the possibility of oppression of an extrapericardial mass (for example incarcerated hiatal hernia) on the heart, leading to the inability to wean the patient from cardiopulmonary bypass [11]. Unrecognized hernias may lead to hemodynamic instability after cardiac surgery. A case of a male patient with such complications caused by hiatus hernia was described. His condition significantly improved after decompression of the stomach. The authors concluded that in all patients with hiatus hernia, a nasogastric tube should be placed prior to cardiac surgery [12]. Giuricin et al. also presented a case report describing a male patient who underwent cardiac surgery. After the operation, the patient developed gastrointestinal symptoms. Five months later, presence of the intestine inside the thoracic cavity was revealed. The decision was made to perform the second surgery [7].
In conclusion, Morgagni hernias are rarely found during adult patients undergoing cardiac surgery but the presence of such a condition usually requires simultaneous to heart operation repair of the diaphragmatic defect.






