Purpose
Angiosarcoma is a sub-type of soft tissue sarcoma originating from malignant transformation of endothelial cells [1]. Such tumors can arise anywhere in the body but most commonly present as cutaneous disease of the scalp and face [2]. Wide local excision alone is associated with a high-rate of local recurrence and high morbidity related to both treatment and disease recurrence [3-7]. Given the frequently diffuse and multifocal nature of cutaneous angiosarcoma, it is not uncommon for the disease to be unresectable at diagnosis. Systemic therapy is an attractive initial treatment modality and is employed for most angiosarcomas of the scalp and face at our institution. The optimal local treatment for this disease has not been determined and could include surgery, radiation therapy, or both modalities. Given the rarity of scalp and face angiosarcoma, prospective studies showing the benefit of radiation therapy are lacking, but several retrospective studies have demonstrated an improved local recurrence and survival with radiation treatment [6, 8-10].
There are multiple approaches to radiation therapy for face and scalp angiosarcoma, including older mixed lateral photon and electron approaches, modern photon radiotherapy techniques, such as intensity-modulated radiotherapy (IMRT) or volumetric-modulated arc therapy (VMAT) as well as high-dose-rate surface applicator (HDR-SA) brachytherapy [11-17]. In our institutional practice, angiosarcoma of the face and scalp is treated with HDR-SA brachytherapy, but a comprehensive evaluation of clinical outcomes with this treatment approach was not performed. Theoretical advantages of this approach include the ability to conform a dose to often irregular, cutaneous target as well as the frequent ability to escalate a dose by delivering “hot spots” adjacent to the surface applicator, within tumor tissue.
We have recently published a study including dosimetric details of a subset of the patients from the current study who were treated with HDR-SA brachytherapy and had detailed planning information available [18]. The current study was a retrospective evaluation of the clinical characteristics and outcomes for the largest cohort, to our knowledge, of scalp and face angiosarcoma patients treated with HDR-SA brachytherapy, between 2003-2018.
Material and methods
Patients’ selection
We identified 20 consecutive patients with histologically confirmed angiosarcoma of the scalp or face that were treated at our institution between 2003-2018 with HDR-SA brachytherapy. With institutional review board approval, these patients were extracted from institutional databases for subsequent review and analysis. All patient’s medical records were reviewed after obtaining approval from our institutional review board.
Therapeutic approach
Our institutional practice in the management of scalp and face angiosarcoma has evolved over the 15 years included in this study, such that there was variability in the use of systemic therapy and surgery, but all patients in the cohort received HDR-SA brachytherapy. For HDR-SA planning, computed tomography (CT) simulation was performed to treat the visualized tumor, with an addition of 5 cm radial clinical margin whenever anatomically feasible. Further technical details of HDR-SA brachytherapy planning have been described [18]. All, except one patient (treated in 2003), were prescribed 51 Gy in 17 fractions. This first patient was prescribed 51.2 Gy in 16 fractions.
Clinical outcomes
Electronic medical records of all patients were reviewed to assess clinical outcomes and treatment-associated toxicity. The two primary acute toxicities studied were dermatitis and alopecia, which were graded as per CTCAE v4.0. Local control (LC) was defined as absence of recurrence within the radiation field or at the margin (≤ 2 cm of the field edge). Local-regional control (LRC) was defined as absence of local recurrence or discontinuous cutaneous regional recurrence (> 2 cm from the field edge) or lymph node recurrence in the head and neck. Distant metastasis was defined as recurrence outside the head and neck area.
Statistical analyses
Time to event calculations were defined as time from start of HDR-SA brachytherapy to the relevant endpoints, and analyzed by Kaplan-Meier method. Event-free survival (EFS) was specified as time from start of HDR-SA brachytherapy to local-regional recurrence (LRR), distant metastasis (DM), or death. Overall survival was defined as time from start of HDR-SA brachytherapy to death or date last known alive. Patients without an event were censored at the date of last follow-up. The log-rank test was used to compare time from HDR-SA brachytherapy to an applicable event. Finally, univariate analyses were performed to determine potential predictors of outcomes (LRC, DM-free survival, and OS). Multivariate analyses were not performed due to small number of patients. A p-value of 0.05 was considered statistically significant for our analyses.
Results
Patient, disease, and pre-brachytherapy treatment characteristics
Characteristics of the 20 patients included in this study are described in Table 1. Angiosarcoma of the scalp or face was initially diagnosed for all patients between 2003-2017. The median age was 66 years (range, 45-85 years) and 15 of 20 patients (75%) were male. The scalp was the primary site of disease in 15 of 20 cases (75%), with multifocal disease being present in 10 of 20 patients (50%).
Table 1
Patient, disease, and pre-brachytherapy treatment characteristics
Sixteen patients received HDR-SA brachytherapy as a part of their initial definitive cancer treatment, while four patients received brachytherapy at the time of disease recurrence or progression. Of patients who had brachytherapy as a part of their initial therapeutic strategy, 7 patients initially underwent surgical resection (treated between 2003 and 2012), 7 patients initially received chemotherapy (treated between 2008 and 2018, most commonly with a taxane-containing regimen), one patient received both surgery and chemotherapy before brachytherapy, and one patient had no previous treatment other than brachytherapy.
Four patients received brachytherapy for recurrent/progressive disease, 1-7 years after other therapy. Two of these patients had progressed 1-2 years after chemotherapy alone and received brachytherapy at that time. In another two patients, brachytherapy was applied for in-field recurrences at 3 and 5 years after initial diagnosis. Both had undergone previous definitive surgery and radiation therapy, with radiation doses of 70.4 Gy (50.4 Gy followed by a 20 Gy boost with IMRT) and 61.2 Gy (with electrons), respectively. Prior to brachytherapy treatment, 3 patients had known lymph node involvement and 3 patients had known distant metastases (to lung or bone).
Clinical outcomes
Four patients did not complete the prescribed HDR-SA brachytherapy course. Two patients interrupted the treatment early (at fraction 14 and 16, for a total dose of 42 Gy and 48 Gy, respectively) due to skin toxicity. Two additional patients discontinued treatment in the context of failure to thrive (at fraction 5 and 7, for a total dose on 15 Gy and 21 Gy, respectively). Of note, the two patients who did not complete the prescribed treatment due to failure to thrive had the largest number of catheters, representing the largest involved disease fields (69 and 49, respectively).
The median follow-up from the start of brachytherapy was 45 months (range, 1-125 months). Clinical outcomes of this patients’ cohort are presented in Table 2. Eight patients had a local disease recurrence (4 in-field and 4 at the margin). The 4-year rate of local control was 63% (95% CI: 45-91%) (Figure 1A). Three patients recurred in the neighboring area ≥ 2 cm outside the field, and 3 patients recurred in lymph nodes only (parotid and/or neck). An additional 4 patients had both cutaneous and nodal recurrence (2 in-field, 1 marginal, and 1 out of field). Median time from the start of brachytherapy to LRR was 19 months (range, 1-60 months), with a 4-year local-regional control rate of 31% (95% CI: 11-53%) (Figure 1B). Median progression-free survival (PFS) from the start of brachytherapy was 16.5 months, with a 4-year PFS of 20% (95% CI: 1-33%) (Figure 1C).
Table 2
Clinical outcomes and salvage
Fig. 1
Patient survival outcomes over time. A) Local control from the start of brachytherapy treatment. B) Local-regional control from the start of brachytherapy treatment. C) Progression-free survival of angiosarcoma patients from the start of brachytherapy treatment. D) Overall survival of angiosarcoma patients from the start of brachytherapy treatment
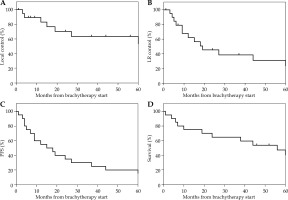
Of the 16 patients with any disease recurrence or progression, 2 had no further therapy, 11 received chemotherapy (5 cases with additional radiation), and 3 patients received a second course of radiation therapy. Repeated brachytherapy alone was applied in 2 patients with marginal field recurrences, and after chemotherapy in 2 patients with out-of-field recurrences. The second course of brachytherapy was also prescribed to 51 Gy in 17 fractions. At the last follow-up, 2 patients were alive, and 2 patients had died with no evidence of recurrence. The median OS was 54 months, and 4-year OS was 54% (95% CI: 35-78%) (Figure 1C).
For the 18 patients with acute skin toxicity, 78% (n = 14) had grade 3 dermatitis, and 22% (n = 4) presented with grade 2 dermatitis. Moist desquamation was managed by cleaning the area with saline compresses, and using aquaphor mixed with 2% lidocaine jelly and/or xeroform gauze dressings. Ibuprofen 200-600 mg every 4-6 hours and/or tylenol 500-1,000 mg every 6-8 hours were also applied as needed. For the 10 patients for whom alopecia information was available, all were noted to have grade 1-2 alopecia.
Predictors of outcome
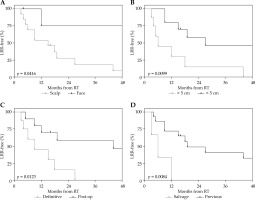
On univariate analysis there was no difference in outcome by sex, focality (unifocal vs. multifocal), or relative timing of chemotherapy (Table 3). As others have shown, a primary anatomic location on the face was associated with better outcomes, including a significantly higher 4-year LRC (75% vs. 9%, p = 0.042; Figure 2A). No patients with primary tumors on the face developed DM at 4 years, while 55% of patients with scalp tumors did so. No association, however, was seen with anatomic site and OS. A smaller tumor size (primary disease < 5 cm) was also associated with significantly better 4-year LRC (35% vs. 0%, p = 0.0099; Figure 2B) and showed a trend towards better 4-year OS (74% vs. 25%, p = 0.06), though had no association with DM. Patients who had surgery prior to HDR-SA brachytherapy (during the same treatment episode) had better outcomes across the board, including higher 4-year LRC (47% vs. 0%, p = 0.0123; Figure 2C), higher rates of being DM-free at 4 years (64% vs. 37%, p = 0.0407), and better 4-year OS (70% vs. 36%, p = 0.0404). HDR-SA brachytherapy as a component of the initial treatment approach also resulted in better 4-year LRC than its use as a component of salvage therapy at the time of disease progression or relapse (32% vs. 0%, p = 0.0084; Figure 2D). This effect was also seen in 4-year OS (66% vs. 0%, p = 0.0019), but no difference was seen in DM rates. Due to small number of patients in this study, multivariate models were not constructed.
Table 3
Univariate predictors of outcome
Fig. 2
The effect of disease and treatment variables on loco-regional recurrence. A) Loco-regional recurrence by anatomic location (scalp vs. face). B) Loco-regional recurrence by tumor size (> 5 cm vs. < 5 cm). C) Loco-regional recurrence by use of prior surgery (definitive vs. salvage). D) Loco-regional recurrence by prior disease progression (salvage vs. previous)
Discussion
Angiosarcoma of the scalp and face is a rare disease with poor prognosis and high rates of local and distant recurrence or progression. Our current institutional approach is multimodality therapy, typically starting with neoadjuvant chemotherapy (primarily taxane-based), followed by HDR-SA brachytherapy. For smaller lesions (typically < 2 cm), surgery alone is sometimes considered. However, this practice has evolved over the past two decades and patients treated at our institution can be referred at a variety of timepoints during their treatment course. As a result, our institutional experience provides a unique opportunity to examine outcomes in the context of consistent use of HDR-SA brachytherapy, with some variability in the utilization of surgery and chemotherapy.
Prior to single-institution, retrospective studies have shown a benefit to radiation (typically as adjuvant therapy) with overall high recurrence rates. The UCLA experience found that 5-year disease-free survival was significantly higher in patients receiving surgery, followed by radiation vs. surgery alone (43% vs. 13%). Five-year actuarial local control was 54% in patients who received adjuvant radiation vs. 19% in patients who had surgery alone [8]. The University of Michigan experience also showed an improved median OS in patients receiving surgery, followed by radiation vs. surgery alone (36.1 months vs. 9.2 months). Local recurrence patterns were not reported by treatment, but 17 of 29 patients developed local recurrence alone, while 4 additional patients developed local recurrence and distant metastasis [9]. The MD Anderson experience also found that combined modality therapy with surgery, followed by radiation was associated with improved OS, disease-free survival, and local control compared to treatment with either surgery or radiation alone. Specifically, the 3-year local control rate with surgery alone or radiation alone was 30%, while the 3-year local control rate with surgery and radiation was 95% [6]. The British Columbia experience showed a trend to improved local relapse-free survival with surgery, followed by radiation vs. surgery alone (70% vs. 40%) [10]. Clinical outcomes in the current study were similar (with 4-year local control of 63%, 4-year local-regional control of 30%, and 4-year PFS of 20%), suggesting HDR-SA brachytherapy may not have significant efficacy advantages over other external beam approaches [3-7]. These disappointing results demonstrate a need for an intensified treatment approach. Given that the majority of patients developed in-field or marginal recurrences, one approach for patients who are not surgical candidates could be a larger initial external beam radiation therapy approach, followed by a brachytherapy boost to the area at highest risk.
In this cohort of patients, univariate analysis demonstrated several factors associated with outcome, which corroborate results of others. Specifically, disease location on the face (vs. scalp) was associated with better prognosis with a 4-year LRC rate of 75% vs. 9% (p = 0.042). A similar result was seen in an analysis from the University of Toronto, where 5-year LRC for the scalp vs. face was 53% vs. 9% (p = 0.04) [19]. A meta-analysis of 11 studies including 379 patients also found that primary location of the scalp was associated with worse prognosis with a 5-year OS odds ratio (OR) of 3.089 (p < 0.001) [20]. This meta-analysis also found that larger tumor size (≥ 5 cm) was significantly associated with worse 5-year OS (OR = 3.566, p = 0.013), which is similar to the trend found in the current study, with a 4-year OS difference of 74% vs. 25% (p = 0.06) and a significant difference in 4-year LRC of 35% vs. 0% (p = 0.0099) based on size. While surgery prior to HDR-SA brachytherapy was associated with better outcomes in our study, this was likely due to the fact that typically, more favorable lesions were treated with surgery. In particular, definitive HDR-SA brachytherapy alone is more often used for lesions spanning a larger area, which we and others have found associated with worse prognosis. In addition, this study found that salvage HDR-SA brachytherapy was associated with worse prognosis than HDR-SA brachytherapy as a component of therapy at initial diagnosis. This difference likely represents the particularly challenging nature of managing progressive or recurrent angiosarcoma, regardless of therapeutic modalities used.
Limitations of this study include its retrospective nature, prone to selection bias. There was also treatment heterogeneity with respect to the use of surgery and systemic therapy. Lastly, the sample size was small. Despite these limitations, given the rarity of cutaneous angiosarcoma of the face and scalp, the findings are informative.
In conclusion, cutaneous angiosarcoma of the face and scalp treated with HDR-SA brachytherapy was associated with moderate local control rates, which are similar to other radiation treatment techniques used either alone or in combination with surgery. Tumors involving the face performed better than those of the scalp. Progression-free survival was poor, and treatment intensification strategies are needed.



