Systemic isotretinoin remains the most efficacious treatment for severe acne as well as many cases of more moderate disease that are unresponsive to other treatment options. Isotretinoin is administered via oral route in the form of capsules containing several excipients, among others soybean oil. For this reason, in case of food allergy and sensitization to soy, a treatment with isotretinoin remains contraindicated [1, 2].
We present hereby the case of a 20-year-old man with severe acne and a history of allergic reaction (oral allergy syndrome) to peach and apple and no history of rhinoconjunctivitis or asthma. This patient was referred to our Unit by his dermatologist to evaluate the safety of isotretinoin treatment (Isodifa capsules, 20 mg/day) [3].
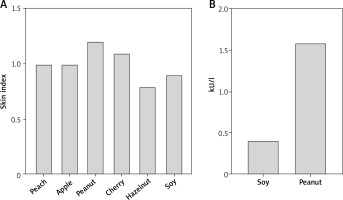
At first, we performed skin testing. A collection of 22 commercial food allergen extracts (Lofarma, Milan, Italy) was used. As for skin reactivity quantitative assessment, the area of the wheals generated was calculated as described [4]. In order to achieve normalization, results were expressed in terms of the ratio between the wheal area and the histamine area, referred to as the Skin Index [4]. As shown in Figure 1 A the skin test was deemed positive for peach (SI = 1), apple (SI = 1), peanut (SI = 1.2), cherry (SI = 1.1), hazelnut (SI = 0.8) and soy (SI = 0.9). Peanut and soy-specific IgEs (ThermoFisher, Italy) were 1.59 and 0.42 KU/l, respectively, with a total IgE value of 198.00 KU/l (Figure 1 B).
Figure 1
Results of the skin testing expressed as skin index (SI) (A); allergen-specific IgE levels in the blood, expressed in kU/l (B)
Using EMBOSS Needle software (EMBL-EBI, Cambridgeshire, UK) we performed the amino acid sequence alignment for soy allergens (Gly m 5, Gly m 6) versus peach (Pru p 3) and apple main allergens (Mal d 1, Mal d 2) seeking for both sequence homology and identity. As shown in Table 1, all the alignments resulted in a very low sequence identity (< 10%), thus ruling out any possible cross-reactivity. Thus, we opted for a single dose oral challenge test rather than an incremental challenge test. Therefore, we administered 1 capsule of Isodifa 20 mg with a subsequent observation for 1 h and 30 min. As expected, the patient tolerated the single dose test and could start and continue the therapy with isotretinoin [5].
Table 1
Amino acid sequence identity between Gly m 5, Gly m 6 and Mal d 1, Mal d 2 and Pru p 3
| Variable | Mal d 1 | Mal d 2 | Pru p 3 |
|---|---|---|---|
| Gly m 5 | 4.7% | 5.9% | 2.2% |
| Gly m 6 | 9.8% | 11.3% | 5.5% |
Here we report the case of a patient with positive skin prick tests and specific IgE for soy and peanut without a history of allergic symptoms to them but with an oral allergy syndrome (OAS) to both peach and apple.
As mentioned before, the treatment with isotretinoin is contraindicated in patients with soy sensitization. According to the literature, in patients with sensitization to soy, an oral challenge test with increasing doses of isotretinoin should be carried out before the treatment is started.
In this case report we demonstrated that in the case of positive skin test/serum IgE to soy in the absence of clinical symptoms, isotretinoin can be introduced in treatment with no adverse reactions.
Moreover, even though the patient had a history of OAS with peach and apple, the low cross-reactivity with the main soy allergens rendered the isotretinoin administration an allow risk procedure [6].
In conclusion, this case report suggests that in the absence of clinical symptoms upon soy ingestion, the treatment with isotretinoin is safe in the patient with positive skin test to soy.








