Purpose
Cervical cancer ranks among the top ten most prevalent gynecologic malignancies in developing countries. Despite improvements in local control and overall survival rates, these outcomes are heavily influenced by the technology and resources available in medical institutions. Unfortunately, in many developing countries, there is a significant disparity in technology between institutions and cities, or even a complete lack of advanced technology.
Brachytherapy, in particular, is highly dependent on technological advancements. While much of its efficacy is not solely due to radiation delivery, technology can greatly enhance this aspect. The ability to quickly assess volumes allows for better dose shaping, and delivering more radiation to the tumor while sparing critical organs, thereby improving overall outcomes. This improvement is well-documented in studies by the EMBRACE group and others. Specifically, volume shaping appears to be a significant game changer.
Magnetic resonance imaging (MRI) stands as the gold standard and state-of-the-art technique for volume-based brachytherapy. Its superior tissue contrast allows for precise delineation of residual tumor and high-risk clinical target volume (HR-CTV). While computed tomography (CT) is also used for volume contouring, it offers less tissue contrast, and comes with its own set of usage guidelines [2].
Ultrasound (US) presents a viable alternative for volume contouring with tissue contrast. Over the years, ultrasound technology has significantly advanced, and currently providing high-quality tissue contrast with the right settings. Furthermore, it can generate volumetric data, given that axial ultrasound slices are correctly spaced, as detailed in our previous work [3]. Although some systems offer real-time 3D ultrasound imaging, their high cost remains a barrier.
Even though ultrasound enables volume contouring, questions remain about the correspondence of these volumes with those obtained from other imaging modalities. This led to a volumetric and dosimetric comparison between CT-acquired and trans-abdominal ultrasound (TAUS)-acquired image sets in brachytherapy applications for cervical cancer.
Material and methods
Data were collected from 18 brachytherapy applications, involving patients with cervical cancer stages IIB-IIIC1. All of them received 3D external-beam radiation therapy (EBRT) with a total dose of 45 Gy/25 fractions. After the 10th session, the start of 3D image-guided brachytherapy with CT was considered, interdigitating applications and EBRT sessions, according to institutional protocol. After a pelvic examination that showed tumor shrinking to manageable volume, brachytherapy applications were scheduled. The procedure was as follows:
Application insertion: Standard insertion was performed, with the addition of a rectal flexible tube and 40 cc of barium gel.
Bladder preparation: A Foley catheter was inserted into the bladder, with the balloon filled with a mixture of 6.5 cc saline and 0.5 cc iodine contrast. The bladder was then emptied and refilled with 250 cc of saline.
Imaging: Orthogonal X-rays were obtained, and the patient was transferred to a CT scanner (Canon Aquilion Prime SP, Canon Medical Systems Corporation, Japan) on a spine board stretcher, secured with straps, and template fixed with Velcro strips.
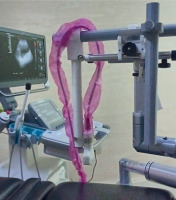
Ultrasound acquisition: Approximately five minutes after CT image, a trans-abdominal ultrasound was performed using a convex 3.5 MHz probe mounted on a 3D-printed arm attached to a brachytherapy stepper (Figure 1), allowing for axial ultrasound slices to be taken every 5 mm. Ultrasound devices used were Toshiba SSA-4000 (Canon Medical Systems Corporation, Tokyo, Japan) and later, Siemens Acuson NX3 Elite (Siemens Healthineers, Erlangen, Germany). Both imaging systems underwent a commissioning for calibration from the vendor with a certificate. In case of Toshiba ultrasound scanner, a test was made with a phantom scanned in US and CT, to assess its validity. The patient was positioned supine, and minimal abdominal fat was necessary to ensure proper transducer movement on the skin. A preliminary scan was conducted to assess bowel loop movement. For patients experiencing gastrointestinal toxicity, loperamide was administered per protocol to prevent increased bowel movement during insertions. The bladder, filled with saline, provided a stable structure unaffected by bowel movements. The stepper and device were secured to the operating table, while the transducer was fixed to the arm in an axial orientation, allowing for axial imaging. Acquisition began at the midline of pubic symphysis, and proceeded in 5 mm increments. A total of 18-20 images were acquired, ending near or at the level of the navel, covering approximately 9-10 cm along the y-axis. Each slide took an average of 2 seconds, resulting in a total acquisition time of 40 seconds, during which the patient was requested to hold the breath. Images from Toshiba scanner were captured using an EasyCap device, while Acuson images were taken and later downloaded in DCM (DICOM) format.
Due to convex nature of the transducer, field of view (FOV) at lesser depths was reduced. However, with the bladder filled as per protocol, it remained within FOV. Along the y-axis, it was not possible to fully assess the rectum or sigmoid colon, which is a limitation of this technique. Nonetheless, analysis indicated that this limitation did not affect the reporting up to D2cc rectum.
Post-treatment delivery with the CT-based plan, the US image set was loaded into treatment planning system (TPS). The set underwent calibration for optimal registration. The applicator configuration was imported as a template from orthogonal X-ray plan that was used also for the CT image set. The US image set was loaded as a secondary image set. Hyperechogenic line within the uterus was aligned with the upper aspect of tandem. Sagittal and coronal views were applied for alignment, with verification performed in axial views to ensure that the hyperechogenic point representing the tandem was within the applicator template.
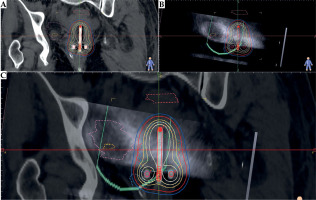
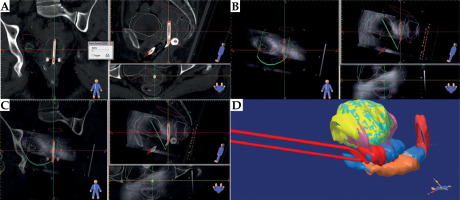
Volume contouring and treatment: Volumes were contoured on CT images, followed by planning and delivery of radiation treatment. The patient was subsequently discharged. Two weeks after the procedure, ultrasound volumes were contoured by the same radiation oncologist, who in most of the cases, contoured CT volumes. TPS used was HDR plus 3.0 (BEBIG Medical, Germany). The upper aspect of the tandem was visible in sagittal-parasagittal images, allowing for alignment with the tandem seen in CT (in original technique, the template is from orthogonal X-rays plan; for the purpose of the study, a template from CT was obtained). Ultrasound volumes (HR-CTV-US, bladder-US, and rectum-US) were contoured. To ensure proper comparison, new bladder-EVAL and rectum-EVAL volumes were created, containing only CT volumes that overlapped with US slices. These volumes were applied for comparison with US volumes (Figures 2 and 3). There was no change of dwelling times, nor monitoring points between the CT-based and US-based plans.
Fig. 2
Details of one of the cases contouring and planning in A) only CT images, B) ultrasound images, and C) a fusion of the two. D) 3D view of the volumes overlapped
Boolean operations were employed to create intersection volumes (planning target volume [PTV]-intersect, bladder-intersect, and rectum-intersect). Subsequently, Dice and Jaccard coefficients were calculated. Descriptive statistics and non-parametric Wilcoxon test were performed on each pair of CT-US volumes. For dose estimation, doses to D90 HR-CTV-US, D2cc bladder-US, and D2cc rectum-US in the administered plan were assessed. Central statistics were used to ensure deviations of less than 5%, and non-parametric Wilcoxon test was conducted to determine whether the means of the doses were equal. Data assessment was performed using GNU PSPP statistical analysis software.
Results
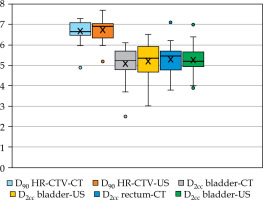
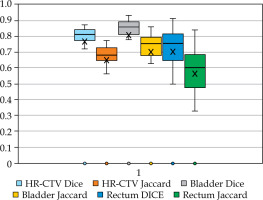
Data from 18 brachytherapy insertions were collected, including 17 with tandem and ring, and 1 with tandem and cylinder. The mean doses to D90 HR-CTV, D2cc rectum, and D2cc bladder for both CT and US are summarized in Table 1 and Figure 4. The mean difference between D90 HR-CTV-CT and D90 HR-CTV-US was 4.3%, below the acceptable difference of 5%. For bladder-CT and bladder-US, the difference was 5.25%, while for rectum-CT and rectum-US, it was 3.98%. Wilcoxon test showed no significant differences between the means of these values (p = 0.291 for HR-CTV, p = 0.154 for bladder, and p = 0.774 for rectum). Regarding volumes, the means for HR-CTV-CT, HR-CTV-US, bladder-CT, and bladder-US are shown in Table 2. Wilcoxon test applied to both HR-CTV volumes indicated a significant difference (p = 0.012), with HR-CTV-US being the lower volume. This finding aligns with other studies, suggesting that CT tends to overestimate the volumes [4]. The mean volumes of bladder-CT and bladder-US were not significantly different (p = 0.557), the same as the mean volumes of rectum-CT and rectum-US (p = 0.078). The means of the Dice and Jaccard coefficients for both sets of volumes are summarized in Table 3 and Figure 5. The bladder volumes showed better correlation than HR-CTV volumes, while the rectum volumes showed the lowest correlation using both Dice and Jaccard coefficients; nevertheless, all results were satisfactory.
Table 1
Mean values and differences between CT and US volumes doses
Table 2
Mean values of CT and US volumes in cc
Discussion
This study aimed to compare CT and TAUS volumes for brachytherapy dose prescription. The technique for acquiring US images was described in a previous study [3]. The use of ultrasound for brachytherapy planning is not new; it has been used since the 1970s [5] and 1980s [6]. Advances in technology, such as B-mode, have made real-time assessment possible, although operator dependency remains a significant issue for volume acquisition.
Ultrasound has been used as 2D images, leveraging its excellent tissue contrast. Dr. Sylvia Van Dyk and her team published their technique [7], validated their measurements against MRI [8], and later reported patient outcomes [9], which were superior to standard planning. The feasibility of ultrasound-based volumes in radiation oncology is well-documented in prostate brachytherapy, which may explain the interest for ultrasound-based volumes in gynecological brachytherapy using trans-rectal ultrasound (TRUS). One study [10] compared TRUS HR-CTV volumes with MRI (with and without applicator) and CT, concluding that TRUS had better correlation than CT. Another study compared a combination of CT and TRUS images [11] with CT alone, both compared with MRI, assuming that the combination was more similar to MRI. These studies also noted that TRUS’s poor ability to contour OARs is a major drawback, mainly due to the higher frequency probes used, which offer more resolution but less depth penetration. The logical next step is to explore a technique that provides information on OARs, which is where TAUS proves its efficacy. HR-CTV volumes in CT and US are very similar, with good to very good Dice [12] and Jaccard coefficients. This similarity is crucial because small variations in volumes, such as D90 HR-CTV or rectum, can lead to significant volume and dose shifts. The correlation in bladder and rectum volumes is less strong but still good, possibly due to the time elapsed between CT and US image acquisition, intra-observer variation, and image registration process.
In some studies, using TRUS, the rectum cannot be assessed as an OAR due to the probe’s nature, position, and limited depth penetration of high-frequency diagnostic ultrasound. A study [14] addressed this issue by defining a “risk zone” in the bladder and rectum (1 cm isometric margin from HR-CTV), achieving good dosimetric results. The assumption that this risk zone is similar in CT and US can explain why their 2 cc doses are similar to that of the current results.
Comparing D90 HR-CTV, 2 cc bladder, and 2 cc rectum in CT and TAUS contours showed no statistical difference. This finding is consistent with studies correlating MRI, CT, and CT-/TRUS-contoured HR-CTV, which showed good correlation. Comparing TAUS-CT or TAUS-MRI also yielded good results. Comparisons between TAUS and other imaging modalities are limited. Van Dyk et al. published their results and experience in various papers, comparing measurements in sagittal and axial TAUS images with corresponding MRI images. A drawback is that TAUS-guided brachytherapy is closer to 2D brachytherapy than volumetric brachytherapy. Nevertheless, their published results [9] are acceptable and support the routine use of TAUS at the Peter MacCallum Cancer Center. The experience of Mahantshetty et al. [15] in Mumbai is similar to the Australian experience, reporting similar results. St-Amant et al. [16] compared CT + 3D-TAUS (with a prototype derived from Clarity Autoscan) with MRI and CT alone. Each imaging modality had an optimized plan, and all were acceptable, but target coverage was best in MRI, followed by 3D-TAUS and CT. Plans of MRI applied to other imaging modalities had less Gy difference for 3D-TAUS + CT than for CT alone, indicating that CT volumes differed from MRI, which could explain the variance in contour volumes. There are some issues regarding the current study. Most of the US contours were done by the same radiation oncologist, although two weeks apart, which could be considered a potential bias. Further advance into this technique requires CT and US contouring by two different researches, which can ensure a true blindness in contouring.
Regarding dose, we aimed at a dose > 80 Gy, according to the first EMBRACE study, in the acknowledgment of starting our clinical experience with 3D brachytherapy. If we assess OARs, the dose to the bladder is kept always below the limit (90 Gy). In the case of 2 cc rectum, the mean dose is higher than the recommended dose to keep the total dose acceptable (4.9 Gy each for 4 applications, following 45 Gy/25 fractions), but the range is 3.8-7.10. In some patients (i.e., the first patient treated, or patients with HR-CTV volume more than 40 cc), the total dose to the rectum was more than 75 Gy, because there was no way of reaching D90 HR-CTV goal and proper constraint to 2 cc rectum; therefore, at physician discretion, reaching D90 HR-CTV goal dose was the choice.
This technique cannot define adequately some OARs, such as the sigmoid and bowel, as they are. This issue remains unresolved, and requires improvement in order to obtain and report a 2 cc sigmoid dose.
It is not possible to define all applicators arrangement from US images. In the original study describing the technique, a previous step was acquiring orthogonal X-ray images using pre-loaded templates of applicators, and arranging them to match the ones in the images. This applicators arrangement can be saved as a template and later loaded into US images set; then, the physicist matched the upper aspect of the tandem with the hyperechogenic line in the uterus in sagittal reconstruction, representing the upper aspect of the tandem (as an hyperechogenic line, with reverberation artifacts). In this study, X-ray images were acquired, the applicators’ templates were created and loaded into the CT images set, while the US images were loaded as a secondary set. The applicators templates were fixed, and the secondary set was aligned making the upper aspect of the tandem in the template, and the hyperechogenic line in the sagittal plane of the US images set was matched.
The use of alternative imaging techniques is justified by the cost of state-of-the-art technologies. Among the available techniques, volumetric ultrasound is a feasible option that warrants further development and implementation.
Conclusions
According to the current study, HR-CTV, bladder, and rectum volumes contoured in both CT and TAUS show good correlation. In a given plan, their doses are similar within the established limit, and close to it in the case of the bladder. The use of ultrasound as an imaging and volumetric reconstruction method is feasible for planning in gynecologic brachytherapy, and its development should be encouraged, especially in regions that cannot afford commonly used imaging methods. More research is warranted, ensuring blindness of the contouring, proper and standard US images acquisition, applicator reconstruction, and complying with GEC-ESTRO recommendations.